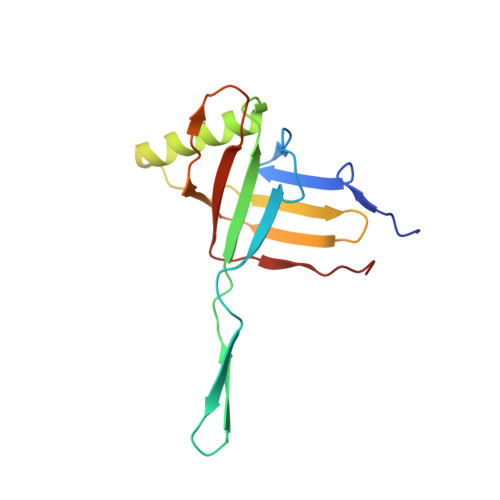
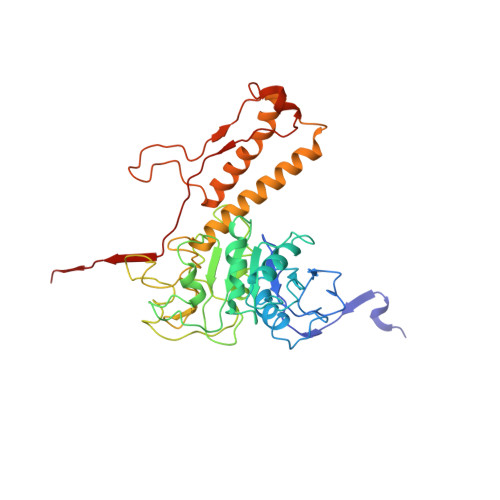
Fine structure and assembly pattern of a minimal myophage Pam3.
Yang, F., Jiang, Y.L., Zhang, J.T., Zhu, J., Du, K., Yu, R.C., Wei, Z.L., Kong, W.W., Cui, N., Li, W.F., Chen, Y., Li, Q., Zhou, C.Z.(2023) Proc Natl Acad Sci U S A 120: e2213727120-e2213727120
- PubMed: 36656854 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.2213727120
- Primary Citation Related Structures:
7YFW, 7YFZ, 8HDR, 8HDS, 8HDT, 8HDW - PubMed Abstract:
The myophage possesses a contractile tail that penetrates its host cell envelope. Except for investigations on the bacteriophage T4 with a rather complicated structure, the assembly pattern and tail contraction mechanism of myophage remain largely unknown. Here, we present the fine structure of a freshwater Myoviridae cyanophage Pam3, which has an icosahedral capsid of ~680 Å in diameter, connected via a three-section neck to an 840-Å-long contractile tail, ending with a three-module baseplate composed of only six protein components. This simplified baseplate consists of a central hub-spike surrounded by six wedge heterotriplexes, to which twelve tail fibers are covalently attached via disulfide bonds in alternating upward and downward configurations. In vitro reduction assays revealed a putative redox-dependent mechanism of baseplate assembly and tail sheath contraction. These findings establish a minimal myophage that might become a user-friendly chassis phage in synthetic biology.
- School of Life Sciences, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230026, China.
Organizational Affiliation: