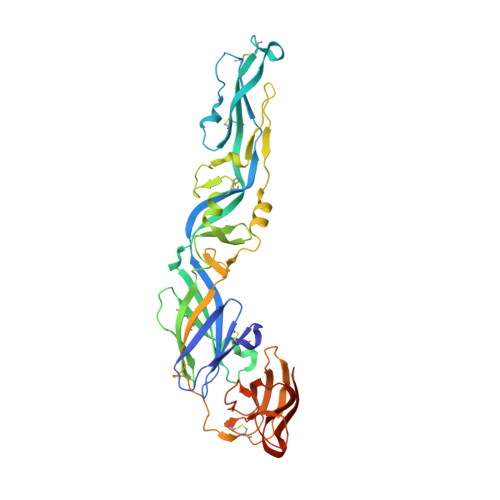
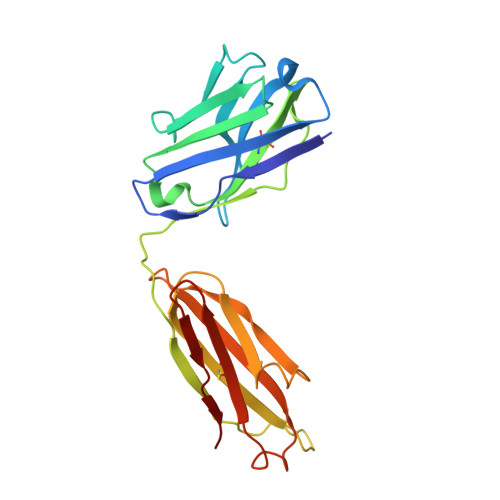
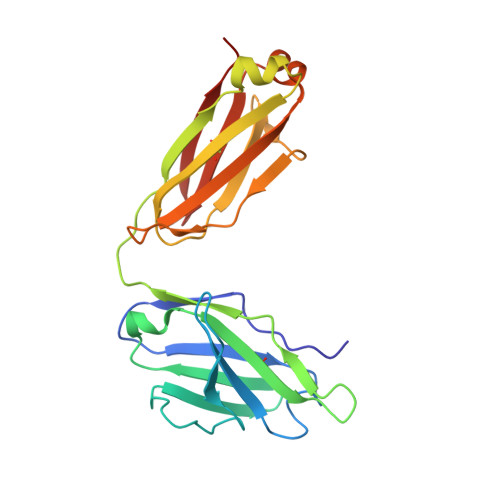
A neutralizing-protective supersite of human monoclonal antibodies for yellow fever virus.
Li, Y., Chen, Z., Wu, L., Dai, L., Qi, J., Chai, Y., Li, S., Wang, Q., Tong, Z., Ma, S., Duan, X., Ren, S., Song, R., Liang, M., Liu, W., Yan, J., Gao, G.F.(2022) Innovation (Camb) 3: 100323-100323
- PubMed: 36199277 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.xinn.2022.100323
- Primary Citation Related Structures:
8GPT, 8GPU, 8GPX - PubMed Abstract:
The yellow fever virus (YFV) is a life-threatening human pathogen. Owing to the lack of available therapeutics, non-vaccinated individuals are at risk. Here, we isolated eight human monoclonal antibodies that neutralize YFV infection. Five recognized overlapping epitopes and exhibited potent neutralizing activity. Two (YD6 and YD73) were ultra-potent and conferred complete protection against the lethal challenge of YFV as both prophylactics and therapeutics in a mouse model. Crystal structures revealed that YD6 engaged the YFV envelope protein in both pre- and post-fusion states, suggesting viral inhibition by a "double-lock" mechanism. The recognition determinants for YD6 and YD73 are clustered at the premembrane (prM)-binding site. Notably, antibodies targeting this site were present in minute traces in YFV-infected individuals but contributed significantly to neutralization, suggesting a vulnerable supersite of YFV. We provide two promising candidates for immunotherapy against YFV, and the supersite represents an ideal target for epitope-based vaccine design.
- CAS Key Laboratory of Pathogen Microbiology and Immunology, Institute of Microbiology, Chinese Academy of Sciences, Beijing 100101, China.
Organizational Affiliation: