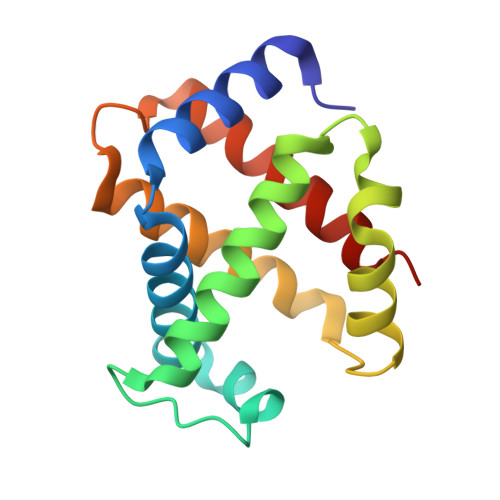
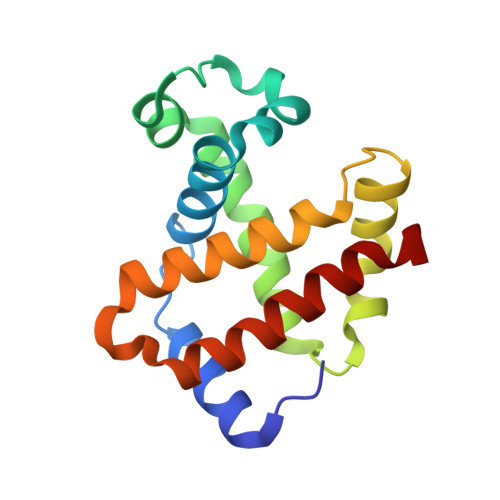
Crystal structural investigations of heme protein derivatives resulting from reactions of aryl- and alkylhydroxylamines with human hemoglobin.
Powell, S.M., Wang, B., Herrera, V.E., Prather, K.Y., Nguyen, N.T., Abucayon, E.G., Thomas, L.M., Safo, M.K., Richter-Addo, G.B.(2023) J Inorg Biochem 246: 112304-112304
- PubMed: 37406385
- DOI: https://doi.org/10.1016/j.jinorgbio.2023.112304
- Primary Citation of Related Structures:
8FDJ, 8FDK, 8FDL, 8FDM, 8FDN - PubMed Abstract:
Phenylhydroxylamine (PhNHOH) and nitrosobenzene (PhNO) interact with human tetrameric hemoglobin (Hb) to form the nitrosobenzene adduct Hb(PhNO). These interactions also frequently lead to methemoglobin formation in red blood cells. We utilize UV-vis spectroscopy and X-ray crystallography to identify the primary and secondary products that form when PhNHOH and related alkylhydroxylamines (RNHOH; R = Me, t-Bu) react with human ferric Hb. We show that with MeNHOH, the primary product is Hb[α-Fe III (H 2 O)][β-Fe II (MeNO)], in which nitrosomethane is bound to the β subunit but not the α subunit. Attempts to isolate a nitrosochloramphenicol (CAMNO) adduct resulted in our isolation of a Hb[α-Fe II ][β-Fe II -cySOx] {CAMNO} product (cySOx = oxidized cysteine) in which CAMNO was located outside of the protein in the solvent region between the β2 and α2 subunits of the same tetramer. We also observed that the βcys93 residue had been oxidized. In the case of t-BuNHOH, we demonstrate that the isolated product is the β-hemichrome Hb[α-Fe III (H 2 O)][β-Fe III (His) 2 ] {t-BuNHOH} , in which the β heme has slipped ∼4.4 Å towards the solvent exterior to accommodate the bis-His heme coordination. When PhNHOH is used, a similar β-hemichrome Hb[α-Fe III (H 2 O)][β-Fe III (His) 2 -cySOx] {PhNHOH} was obtained. Our results reveal, for the first time, the X-ray structural determination of a β-hemichrome in a human Hb derivative. Our UV-vis and X-ray crystal structural result reveal that although Hb(PhNO) and Hb(RNO) complexes may form as primary products, attempted isolation of these products by crystallization may result in the structural determination of their secondary products which may contain β-hemichromes en route to further protein degradation.
- Price Family Foundation Institute of Structural Biology, Department of Chemistry and Biochemistry, University of Oklahoma, 101 Stephenson Parkway, Norman, OK 73019, USA; Biological Sciences Division, Pacific Northwest National Laboratory, Richland, WA 99352, USA.
Organizational Affiliation: