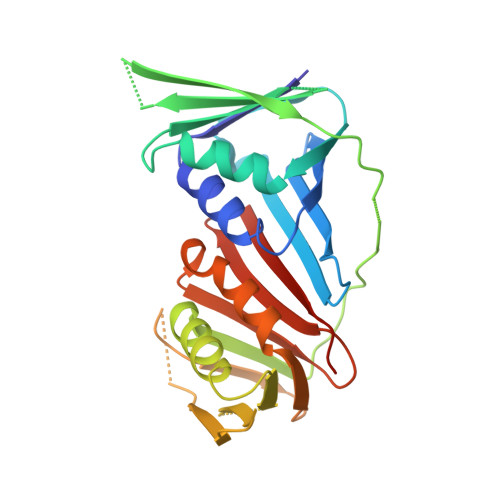
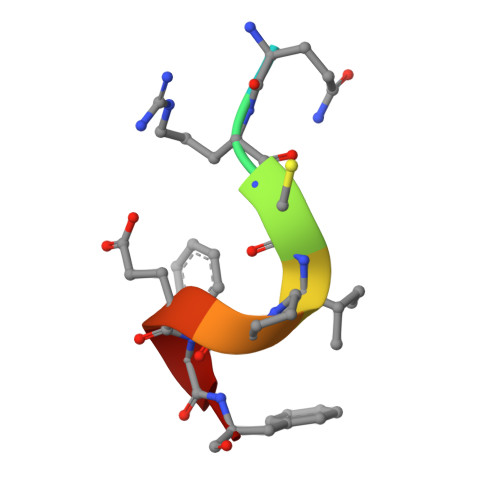
Molecular insight into the PCNA-binding mode of FBH1.
Liu, J., Chaves-Arquero, B., Wei, P., Tencer, A.H., Ruiz-Albor, A., Zhang, G., Blanco, F.J., Kutateladze, T.G.(2023) Structure 31: 511-517.e3
- PubMed: 36990095 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2023.03.004
- Primary Citation Related Structures:
8F5Q - PubMed Abstract:
F-box DNA helicase 1 (FBH1) is involved in the regulation of cell responses to replicative stress. FBH1 is recruited to stalled DNA replication fork by PCNA where it inhibits homologous recombination and catalyzes fork regression. Here, we report the structural basis for the molecular recognition of two distinctly different motifs of FBH1, FBH1 PIP and FBH1 APIM , by PCNA. The crystal structure of PCNA in complex with FBH1 PIP and analysis of NMR perturbations reveal overlapped FBH1 PIP and FBH1 APIM binding sites of PCNA and the dominant contribution of FBH1 PIP in this interaction.
- Department of Pharmacology, University of Colorado School of Medicine, Aurora, CO 80045, USA.
Organizational Affiliation: