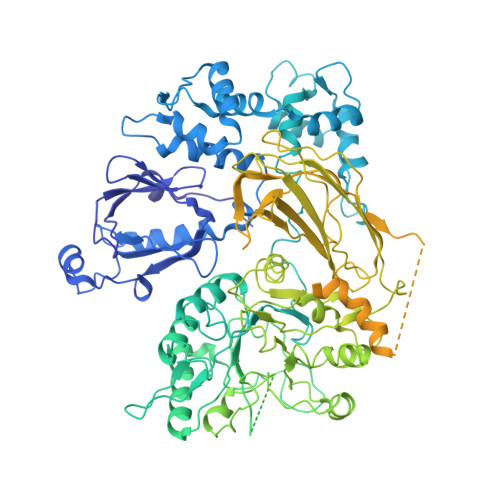
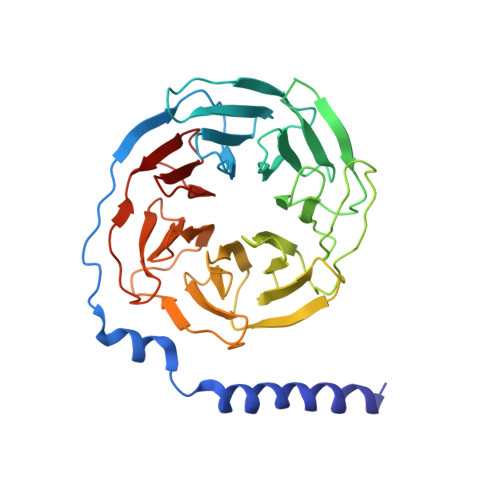
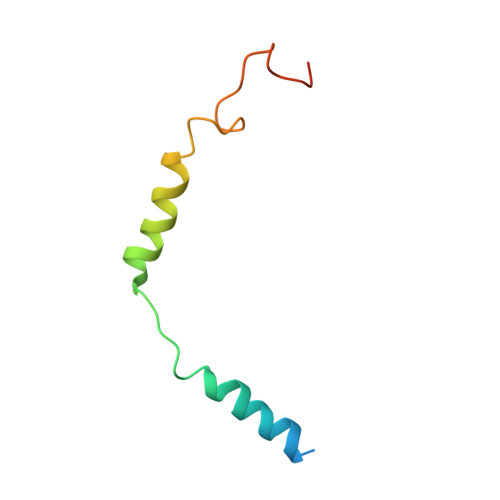
G beta gamma activates PIP2 hydrolysis by recruiting and orienting PLC beta on the membrane surface.
Falzone, M.E., MacKinnon, R.(2023) Proc Natl Acad Sci U S A 120: e2301121120-e2301121120
- PubMed: 37172014 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.2301121120
- Primary Citation Related Structures:
8EMV, 8EMW, 8EMX - PubMed Abstract:
Phospholipase C-βs (PLCβs) catalyze the hydrolysis of phosphatidylinositol 4, 5-bisphosphate [Formula: see text] into [Formula: see text] [Formula: see text] and [Formula: see text] [Formula: see text]. [Formula: see text] regulates the activity of many membrane proteins, while IP3 and DAG lead to increased intracellular Ca 2+ levels and activate protein kinase C, respectively. PLCβs are regulated by G protein-coupled receptors through direct interaction with [Formula: see text] and [Formula: see text] and are aqueous-soluble enzymes that must bind to the cell membrane to act on their lipid substrate. This study addresses the mechanism by which [Formula: see text] activates PLCβ 3. We show that PLCβ 3 functions as a slow Michaelis-Menten enzyme ( [Formula: see text] ) on membrane surfaces. We used membrane partitioning experiments to study the solution-membrane localization equilibrium of PLCβ 3. Its partition coefficient is such that only a small quantity of PLCβ 3 exists in the membrane in the absence of [Formula: see text] . When [Formula: see text] is present, equilibrium binding on the membrane surface increases PLCβ 3 in the membrane, increasing [Formula: see text] in proportion. Atomic structures on membrane vesicle surfaces show that two [Formula: see text] anchor PLCβ 3 with its catalytic site oriented toward the membrane surface. Taken together, the enzyme kinetic, membrane partitioning, and structural data show that [Formula: see text] activates PLCβ by increasing its concentration on the membrane surface and orienting its catalytic core to engage [Formula: see text] . This principle of activation explains rapid stimulated catalysis with low background activity, which is essential to the biological processes mediated by [Formula: see text] , IP3 , and DAG .
- Laboratory of Molecular Neurobiology and Biophysics, The Rockefeller University, New York, NY 10065.
Organizational Affiliation: