Crystal Structure Analysis of xHDMX in complex with the stapled peptide PROTAC analog
Seo, H.-S., Dhe-Paganon, S.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| HDMX | 131 | Danio rerio | Mutation(s): 0 | 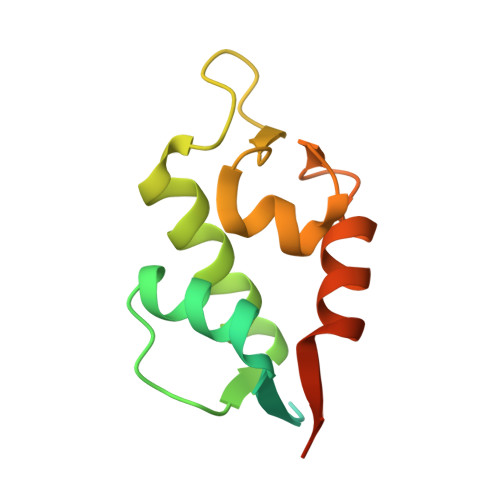 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q7ZUW7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| ACE-LEU-THR-PHE-0EH-GLU-TYR-TRP-ALA-GLN-LEU-MK8-SER-ALA-ALA | 16 | synthetic construct | Mutation(s): 0 | 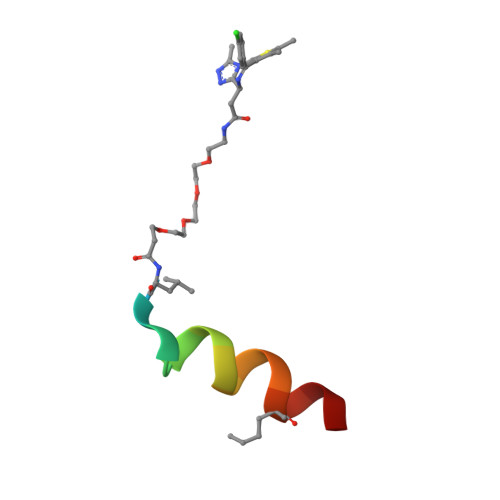 | |
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| 0EH Query on 0EH | B | D-PEPTIDE LINKING | C10 H21 N O2 |  | -- |
| MK8 Query on MK8 | B | L-PEPTIDE LINKING | C7 H15 N O2 |  | LEU |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 81.52 | α = 90 |
| b = 108.4 | β = 90 |
| c = 30.73 | γ = 90 |
| Software Name | Purpose |
|---|---|
| XDS | data reduction |
| xia2 | data scaling |
| PHASER | phasing |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Cancer Institute (NIH/NCI) | United States | -- |