Postfusion Nipah virus fusion protein in complex with Fab 1H1
Byrne, P.O., Blade, E.G., McLellan, J.S.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Fusion glycoprotein F0,Fusion glycoprotein F1 | 529 | Henipavirus nipahense | Mutation(s): 0 | 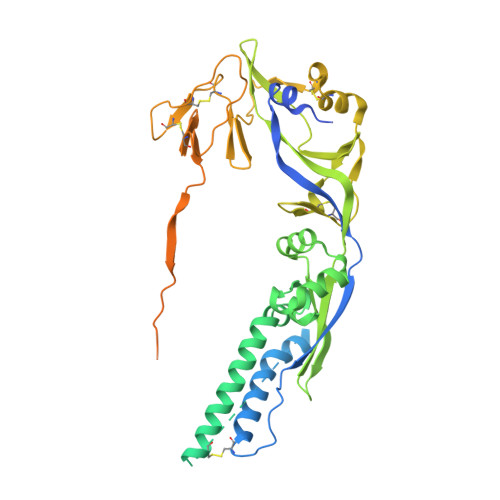 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9IH63 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| antibody 1H1 heavy chain | D, F [auth H] | 121 | Mus musculus | Mutation(s): 0 | 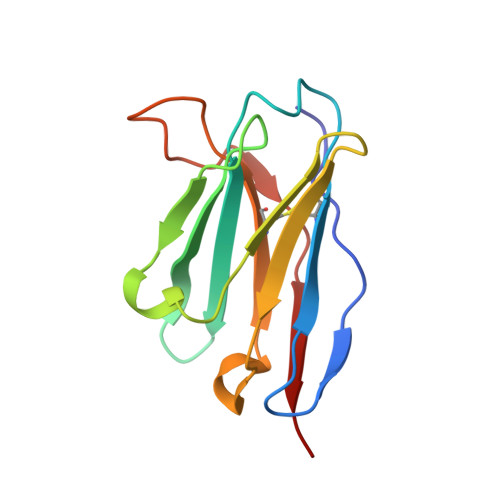 |
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| antibody 1H1 light chain | E, G [auth L] | 107 | Mus musculus | Mutation(s): 0 |  |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Welch Foundation | United States | F-0003-19620604 |