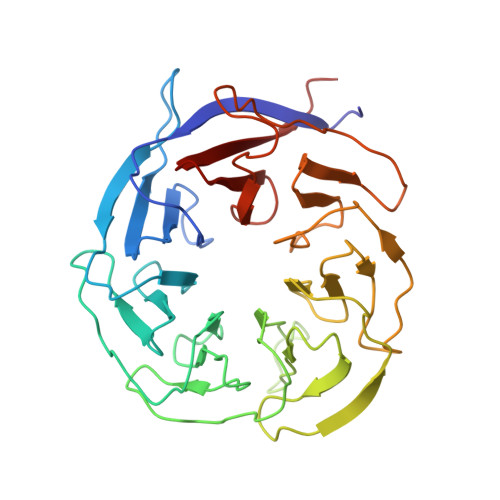
mRNA reading frame maintenance during eukaryotic ribosome translocation.
Milicevic, N., Jenner, L., Myasnikov, A., Yusupov, M., Yusupova, G.(2024) Nature 625: 393-400
- PubMed: 38030725 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-023-06780-4
- Primary Citation Related Structures:
8CCS, 8CDL, 8CDR, 8CEH, 8CF5, 8CG8, 8CGN, 8CIV, 8CKU, 8CMJ - PubMed Abstract:
One of the most critical steps of protein synthesis is coupled translocation of messenger RNA (mRNA) and transfer RNAs (tRNAs) required to advance the mRNA reading frame by one codon. In eukaryotes, translocation is accelerated and its fidelity is maintained by elongation factor 2 (eEF2) 1,2 . At present, only a few snapshots of eukaryotic ribosome translocation have been reported 3-5 . Here we report ten high-resolution cryogenic-electron microscopy (cryo-EM) structures of the elongating eukaryotic ribosome bound to the full translocation module consisting of mRNA, peptidyl-tRNA and deacylated tRNA, seven of which also contained ribosome-bound, naturally modified eEF2. This study recapitulates mRNA-tRNA 2 -growing peptide module progression through the ribosome, from the earliest states of eEF2 translocase accommodation until the very late stages of the process, and shows an intricate network of interactions preventing the slippage of the translational reading frame. We demonstrate how the accuracy of eukaryotic translocation relies on eukaryote-specific elements of the 80S ribosome, eEF2 and tRNAs. Our findings shed light on the mechanism of translation arrest by the anti-fungal eEF2-binding inhibitor, sordarin. We also propose that the sterically constrained environment imposed by diphthamide, a conserved eukaryotic posttranslational modification in eEF2, not only stabilizes correct Watson-Crick codon-anticodon interactions but may also uncover erroneous peptidyl-tRNA, and therefore contribute to higher accuracy of protein synthesis in eukaryotes.
- Institute of Genetics and Molecular and Cellular Biology (IGBMC), CNRS UMR7104, INSERM U1258, University of Strasbourg, Strasbourg, France.
Organizational Affiliation: