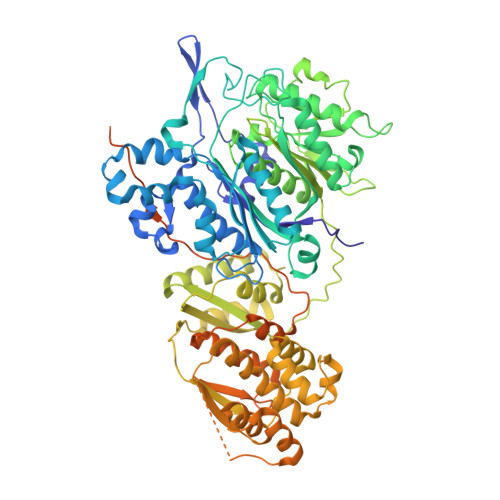
Architecture of a PKS-NRPS hybrid megaenzyme involved in the biosynthesis of the genotoxin colibactin.
Bonhomme, S., Contreras-Martel, C., Dessen, A., Macheboeuf, P.(2023) Structure 31: 700
- PubMed: 37059096 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2023.03.012
- Primary Citation Related Structures:
8CJH - PubMed Abstract:
The genotoxin colibactin produced by Escherichia coli is involved in the development of colorectal cancers. This secondary metabolite is synthesized by a multi-protein machinery, mainly composed of non-ribosomal peptide synthetase (NRPS)/polyketide synthase (PKS) enzymes. In order to decipher the function of a PKS-NRPS hybrid enzyme implicated in a key step of colibactin biosynthesis, we conducted an extensive structural characterization of the ClbK megaenzyme. Here we present the crystal structure of the complete trans-AT PKS module of ClbK showing structural specificities of hybrid enzymes. In addition, we report the SAXS solution structure of the full-length ClbK hybrid that reveals a dimeric organization as well as several catalytic chambers. These results provide a structural framework for the transfer of a colibactin precursor through a PKS-NRPS hybrid enzyme and can pave the way for re-engineering PKS-NRPS hybrid megaenzymes to generate diverse metabolites with many applications.
- University Grenoble Alpes, CNRS, CEA, Institut de Biologie Structurale (IBS), Bacterial Pathogenesis Group, 38000 Grenoble, France.
Organizational Affiliation: