Structure of human WDR5 and pVHL:ElonginC:ElonginB bound to PROTAC with PEG linker (conformation #1)
Kraemer, A., Doelle, A., Knapp, S., Structural Genomics Consortium (SGC)To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Elongin-B | A [auth J] | 104 | Homo sapiens | Mutation(s): 0 Gene Names: ELOB, TCEB2 | 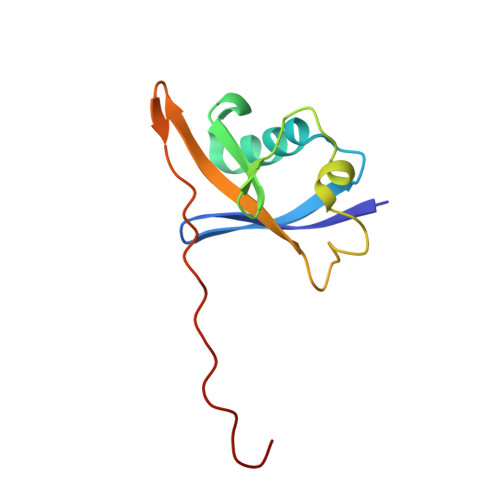 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q15370 GTEx: ENSG00000103363 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q15370 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Elongin-C | B [auth K] | 98 | Homo sapiens | Mutation(s): 0 Gene Names: ELOC, TCEB1 | 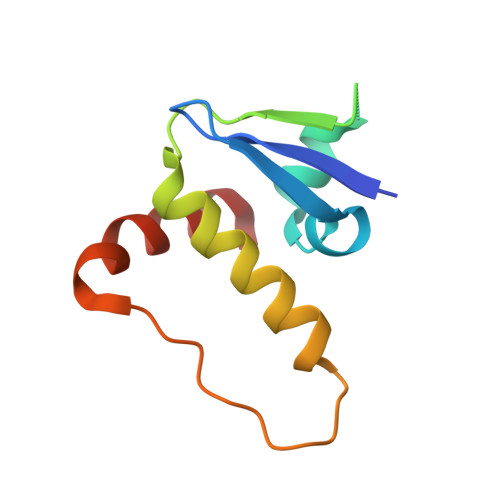 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q15369 GTEx: ENSG00000154582 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q15369 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| von Hippel-Lindau disease tumor suppressor | C [auth L] | 162 | Homo sapiens | Mutation(s): 0 Gene Names: VHL | 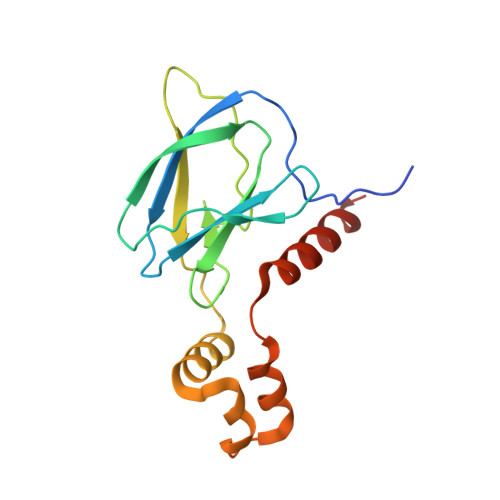 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P40337 GTEx: ENSG00000134086 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P40337 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| WD repeat-containing protein 5 | D [auth B] | 321 | Homo sapiens | Mutation(s): 0 Gene Names: WDR5, BIG3 |  |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P61964 GTEx: ENSG00000196363 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P61964 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| Q3X (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | AA [auth B] | ~{N}-[5-[4-[2-[2-[2-[2-[3-[[3,3-dimethyl-1-[(2~{S},4~{R})-2-[[4-(4-methyl-1,3-thiazol-5-yl)phenyl]methylcarbamoyl]-4-oxidanyl-pyrrolidin-1-yl]-1-oxidanylidene-butan-2-yl]amino]-3-oxidanylidene-propoxy]ethoxy]ethoxy]ethoxy]ethylcarbamoyl]phenyl]-2-(4-methylpiperazin-1-yl)phenyl]-6-oxidanylidene-4-(trifluoromethyl)-3~{H}-pyridine-3-carboxamide C58 H72 F3 N9 O11 S MZOFELXFRDGDBN-GYPJNVOQSA-N |  | ||
| P6G Download:Ideal Coordinates CCD File | J [auth K] | HEXAETHYLENE GLYCOL C12 H26 O7 IIRDTKBZINWQAW-UHFFFAOYSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | BA [auth B] CA [auth B] DA [auth B] EA [auth B] FA [auth B] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| SCN Download:Ideal Coordinates CCD File | E [auth J] F [auth J] G [auth J] K M [auth L] | THIOCYANATE ION C N S ZMZDMBWJUHKJPS-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 47.572 | α = 90 |
| b = 189.858 | β = 117.64 |
| c = 49.723 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| XDS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| PDB_EXTRACT | data extraction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| The Structural Genomics Consortium (SGC) | Canada | -- |