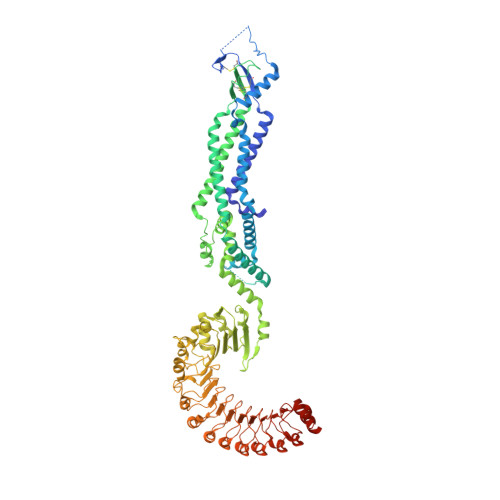
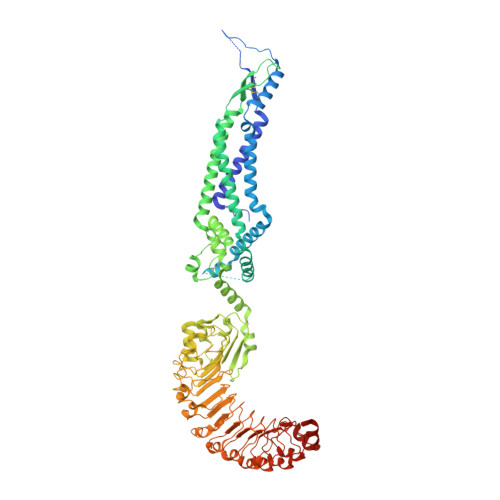
Structure of a volume-regulated heteromeric LRRC8A/C channel.
Rutz, S., Deneka, D., Dittmann, A., Sawicka, M., Dutzler, R.(2023) Nat Struct Mol Biol 30: 52-61
- PubMed: 36522427 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-022-00899-0
- Primary Citation Related Structures:
8B40, 8B41, 8B42, 8BEN - PubMed Abstract:
Volume-regulated anion channels (VRACs) participate in the cellular response to osmotic swelling. These membrane proteins consist of heteromeric assemblies of LRRC8 subunits, whose compositions determine permeation properties. Although structures of the obligatory LRRC8A, also referred to as SWELL1, have previously defined the architecture of VRACs, the organization of heteromeric channels has remained elusive. Here we have addressed this question by the structural characterization of murine LRRC8A/C channels. Like LRRC8A, these proteins assemble as hexamers. Despite 12 possible arrangements, we find a predominant organization with an A:C ratio of two. In this assembly, four LRRC8A subunits cluster in their preferred conformation observed in homomers, as pairs of closely interacting proteins that stabilize a closed state of the channel. In contrast, the two interacting LRRC8C subunits show a larger flexibility, underlining their role in the destabilization of the tightly packed A subunits, thereby enhancing the activation properties of the protein.
- Department of Biochemistry, University of Zurich, Zurich, Switzerland.
Organizational Affiliation: