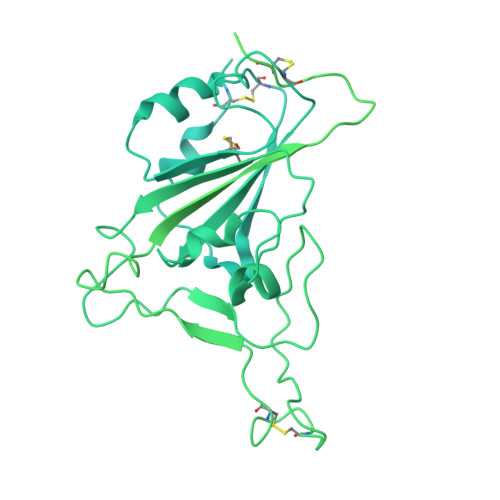
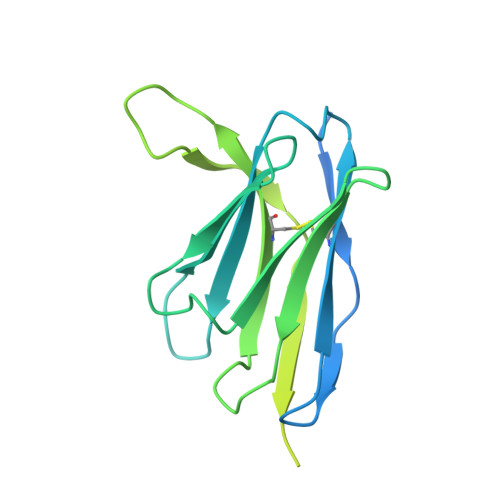
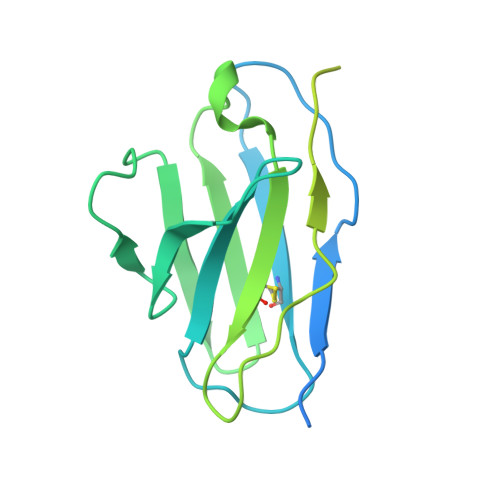
Potent neutralizing broad-spectrum antibody against SARS-CoV-2 generated from dual-antigen-specific B cells from convalescents.
Takeshita, M., Fukuyama, H., Kamada, K., Matsumoto, T., Makino-Okamura, C., Lin, Q., Sakuma, M., Kawahara, E., Yamazaki, I., Uchikubo-Kamo, T., Tomabechi, Y., Hanada, K., Hisano, T., Moriyama, S., Takahashi, Y., Ito, M., Imai, M., Maemura, T., Furusawa, Y., Yamayoshi, S., Kawaoka, Y., Shirouzu, M., Ishii, M., Saya, H., Kondo, Y., Kaneko, Y., Suzuki, K., Fukunaga, K., Takeuchi, T.(2023) iScience 26: 106955-106955
- PubMed: 37288342 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.isci.2023.106955
- Primary Citation Related Structures:
7X93, 7X94, 7X95, 7X96, 7Y6L, 7Y6N - PubMed Abstract:
Several antibody therapeutics have been developed against SARS-CoV-2; however, they have attenuated neutralizing ability against variants. In this study, we generated multiple broadly neutralizing antibodies from B cells of convalescents, by using two types of receptor-binding domains, Wuhan strain and the Gamma variant as bait. From 172 antibodies generated, six antibodies neutralized all strains prior to the Omicron variant, and the five antibodies were able to neutralize some of the Omicron sub-strains. Structural analysis showed that these antibodies have a variety of characteristic binding modes, such as ACE2 mimicry. We subjected a representative antibody to the hamster infection model after introduction of the N297A modification, and observed a dose-dependent reduction of the lung viral titer, even at a dose of 2 mg/kg. These results demonstrated that our antibodies have certain antiviral activity as therapeutics, and highlighted the importance of initial cell-screening strategy for the efficient development of therapeutic antibodies.
- Division of Rheumatology, Department of Internal Medicine, Keio University School of Medicine, Tokyo 160-8582, Japan.
Organizational Affiliation: