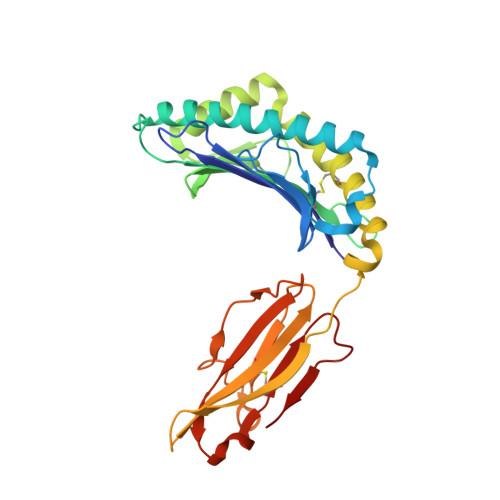
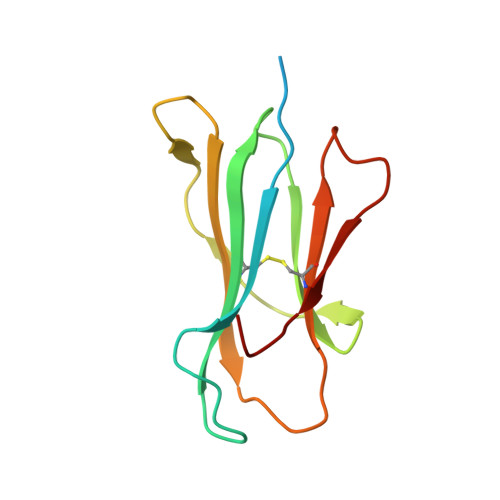
Analysis of the characteristics of feline major histocompatibility complex class I molecules cross-presenting coronavirus peptides
Qiao, P.W., Yue, C., Peng, W.Y., Liu, K.F., Huo, S.T., Zhang, D., Chai, Y., Qi, J.X., Sun, Z.Y., Gao, G.F., Liu, W.J., Wu, G.Z.To be published.