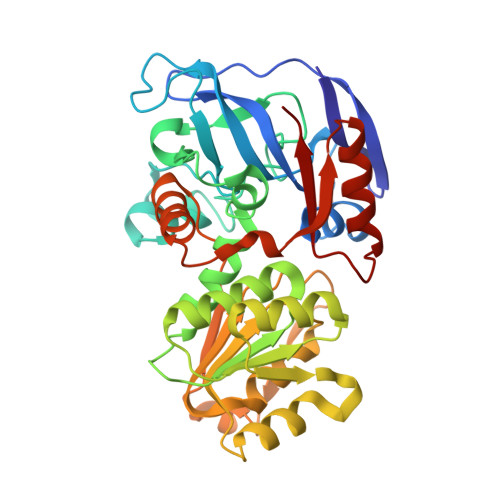
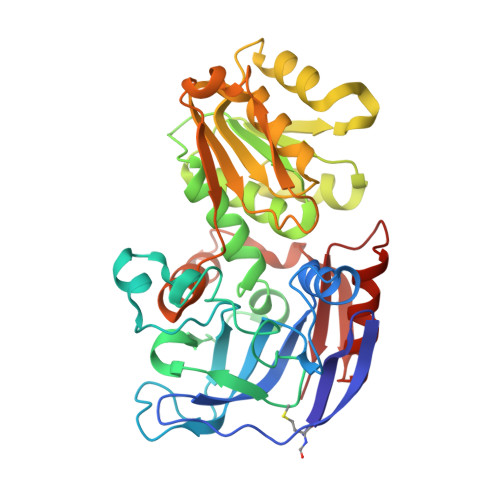
Crystallographic snapshots of ternary complexes of thermophilic secondary alcohol dehydrogenase from Thermoanaerobacter pseudoethanolicus reveal the dynamics of ligand exchange and the proton relay network.
Dinh, T., Rahn, K.T., Phillips, R.S.(2022) Proteins 90: 1570-1583
- PubMed: 35357038
- DOI: https://doi.org/10.1002/prot.26339
- Primary Citation Related Structures:
7UTC, 7UUT, 7UX4 - PubMed Abstract:
Three-dimensional structures of I86A and C295A mutant secondary alcohol dehydrogenase (SADH) from Thermoanaerobacter pseudoethanolicus were determined by x-ray crystallography. The tetrameric structure of C295A-SADH soaked with NADP + and dimethyl sulfoxide (DMSO) was determined to 1.85 Å with an R free of 0.225. DMSO is bound to the tetrahedral zinc in each subunit, with ligands from SG of Cys-37, NE2 of His-59, and OD2 of Asp-150. The nicotinamide ring of NADP is hydrogen-bonded to the N of Ala-295 and the O of Val-265 and Gly-293. The O of DMSO is connected to a network of hydrogen bonds with OG of Ser-39, the 3'-OH of NADP, and ND1 of His-42. The structure of I86A-SADH soaked with 2-pentanol and NADP + contains (R)-2-pentanol bound in each subunit, ligated to the tetrahedral zinc, and connected to the proton relay network. The structure of I86A-SADH soaked with 3-methylcyclohexanol and NADP + has alcohol bound in three subunits. Two of the sites have the alcohol ligated to the zinc in an axial position, with OE2 of Glu-60 in the other axial position of a trigonal bipyramidal complex. One site has 3-methylcyclohexanol bound noncovalently, with the zinc in an inverted tetrahedral geometry with Glu-60. The fourth site also has the zinc in a trigonal bipyramidal complex with axial Glu-60 and water ligands. These structures demonstrate that ligand exchange of SADH involves pentacoordinate and inverted zinc complexes with Glu-60. Furthermore, we see a network of hydrogen bonds connecting the substrate oxygen to the external solvent that is likely to play a role in the mechanism of SADH.
- Department of Chemistry, University of Georgia, Athens, Georgia, USA.
Organizational Affiliation: