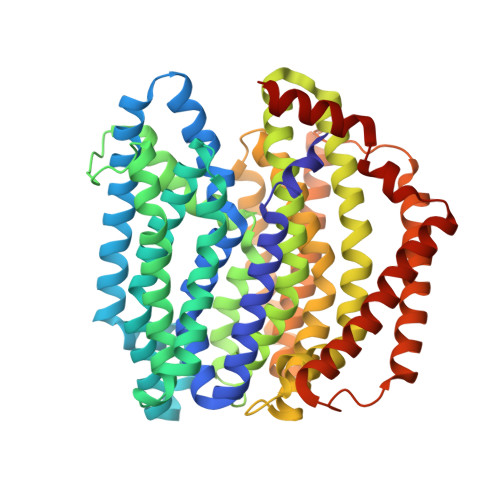
Crystal structure of the lipid flippase MurJ in a "squeezed" form distinct from its inward- and outward-facing forms.
Kohga, H., Mori, T., Tanaka, Y., Yoshikaie, K., Taniguchi, K., Fujimoto, K., Fritz, L., Schneider, T., Tsukazaki, T.(2022) Structure 30: 1088-1097.e3
- PubMed: 35660157 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2022.05.008
- Primary Citation Related Structures:
7WAG, 7WAW, 7WAX - PubMed Abstract:
The bacterial peptidoglycan enclosing the cytoplasmic membrane is a fundamental cellular architecture. The integral membrane protein MurJ plays an essential role in flipping the cell wall building block Lipid II across the cytoplasmic membrane for peptidoglycan biosynthesis. Previously reported crystal structures of MurJ have elucidated its V-shaped inward- or outward-facing forms with an internal cavity for substrate binding. MurJ transports Lipid II using its cavity through conformational transitions between these two forms. Here, we report two crystal structures of inward-facing forms from Arsenophonus endosymbiont MurJ and an unprecedented crystal structure of Escherichia coli MurJ in a "squeezed" form, which lacks a cavity to accommodate the substrate, mainly because of the increased proximity of transmembrane helices 2 and 8. Subsequent molecular dynamics simulations supported the hypothesis that the squeezed form is an intermediate conformation. This study fills a gap in our understanding of the Lipid II flipping mechanism.
- Nara Institute of Science and Technology, Ikoma, Nara 630-0192, Japan.
Organizational Affiliation: