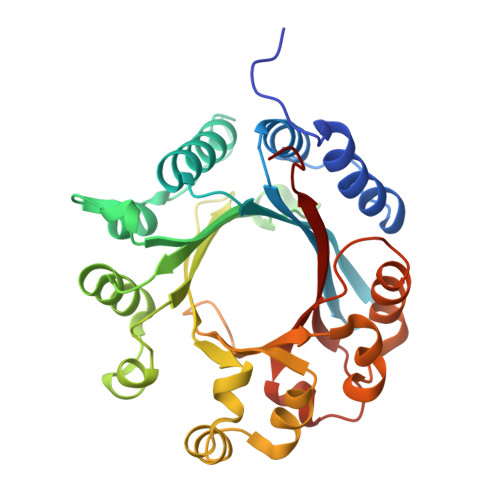
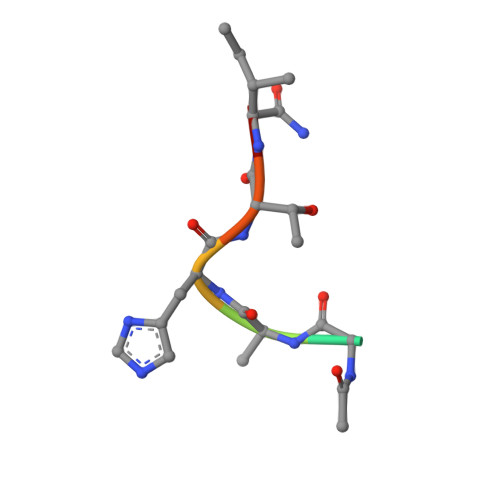
LimF is a versatile prenyltransferase for histidine-C-geranylation on diverse non-natural substrates
Zhang, Y., Hamada, K., Nguyen, D.T., Inoue, S., Satake, M., Kobayashi, S., Okada, C., Ogata, K., Okada, M., Sengoku, T., Goto, Y., Suga, H.(2022) Nat Catal