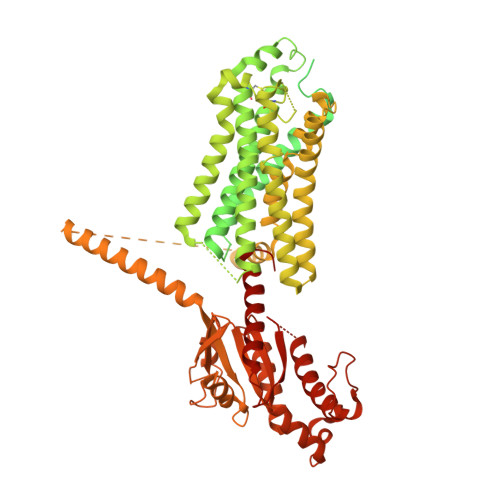
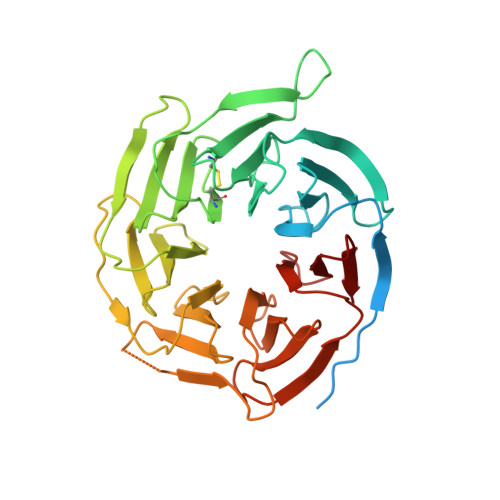
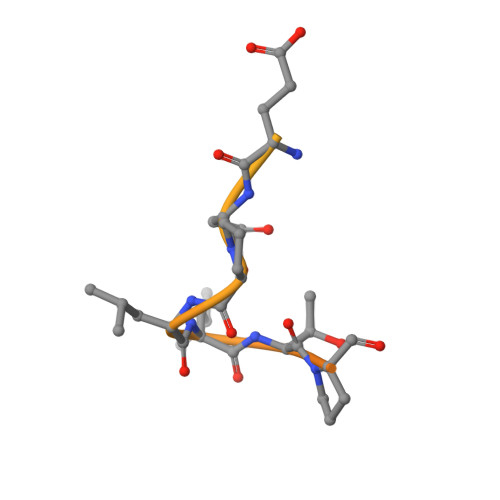
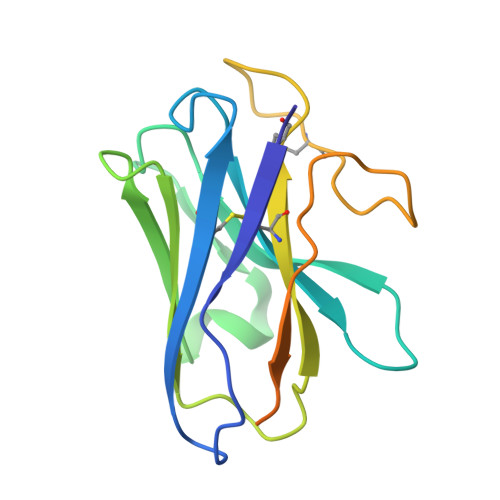
The relaxin receptor RXFP1 signals through a mechanism of autoinhibition.
Erlandson, S.C., Rawson, S., Osei-Owusu, J., Brock, K.P., Liu, X., Paulo, J.A., Mintseris, J., Gygi, S.P., Marks, D.S., Cong, X., Kruse, A.C.(2023) Nat Chem Biol 19: 1013-1021
- PubMed: 37081311 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41589-023-01321-6
- Primary Citation Related Structures:
7TMW - PubMed Abstract:
The relaxin family peptide receptor 1 (RXFP1) is the receptor for relaxin-2, an important regulator of reproductive and cardiovascular physiology. RXFP1 is a multi-domain G protein-coupled receptor (GPCR) with an ectodomain consisting of a low-density lipoprotein receptor class A (LDLa) module and leucine-rich repeats. The mechanism of RXFP1 signal transduction is clearly distinct from that of other GPCRs, but remains very poorly understood. In the present study, we determine the cryo-electron microscopy structure of active-state human RXFP1, bound to a single-chain version of the endogenous agonist relaxin-2 and the heterotrimeric G s protein. Evolutionary coupling analysis and structure-guided functional experiments reveal that RXFP1 signals through a mechanism of autoinhibition. Our results explain how an unusual GPCR family functions, providing a path to rational drug development targeting the relaxin receptors.
- Department of Biological Chemistry and Molecular Pharmacology, Blavatnik Institute, Harvard Medical School, Boston, MA, USA.
Organizational Affiliation: