Crystal structure of Saccharomyces cerevisiae NADH-cytochrome b5 reductase 1 (Cbr1) fragment (residues 28-284) bound to FAD
Fenwick, M.K., Lin, H.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-cytochrome b5 reductase 1 | 261 | Saccharomyces cerevisiae | Mutation(s): 0 Gene Names: CBR1, CBR, CBR5, YIL043C EC: 1.6.2.2 | 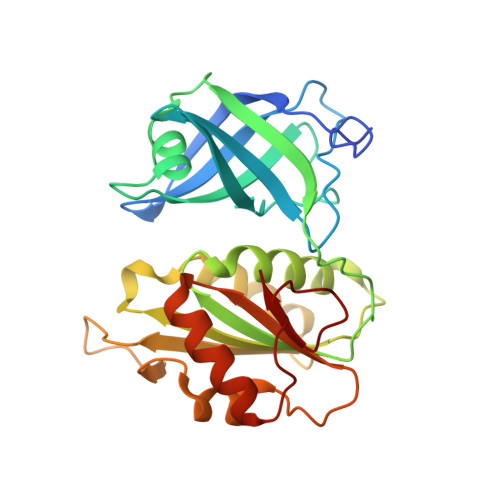 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P38626 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FAD (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | B [auth A] | FLAVIN-ADENINE DINUCLEOTIDE C27 H33 N9 O15 P2 VWWQXMAJTJZDQX-UYBVJOGSSA-N |  | ||
| PGE Download:Ideal Coordinates CCD File | F [auth A], G [auth A] | TRIETHYLENE GLYCOL C6 H14 O4 ZIBGPFATKBEMQZ-UHFFFAOYSA-N |  | ||
| CL Download:Ideal Coordinates CCD File | C [auth A], D [auth A], E [auth A] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 72.225 | α = 90 |
| b = 72.225 | β = 90 |
| c = 114.669 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Aimless | data scaling |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Disease (NIH/NIDDK) | United States | DK107868-04 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | GM124165-01 |
| National Institutes of Health/Office of the Director | United States | S10OD021527 |
| Department of Energy (DOE, United States) | United States | DE-AC02-06CH11357 |