A mutant of the nitrile hydratase from Geobacillus pallidus having enhanced thermostability
Van Wyk, J.C., Cowan, D.A., Danson, M.J., Tsekoa, T.L., Sayed, M.F., Sewell, B.T.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Nitrile hydratase | 216 | Aeribacillus pallidus | Mutation(s): 1 EC: 4.2.1.84 | 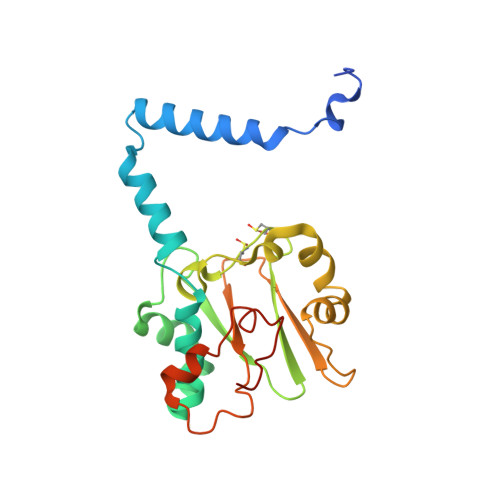 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q84FS5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Nitrile hydratase subunit beta | 229 | Aeribacillus pallidus | Mutation(s): 2 EC: 4.2.1.84 | 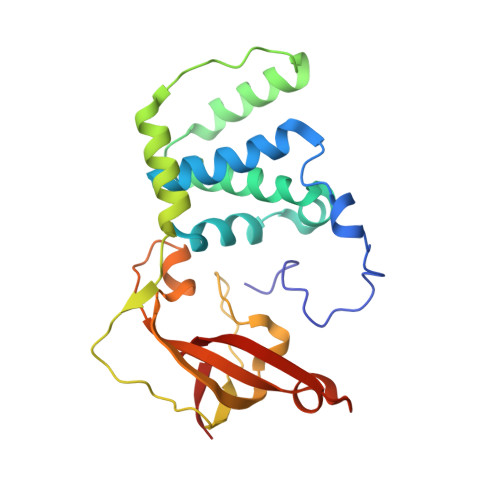 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q84FS6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 3CO (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth A] | COBALT (III) ION Co JAWGVVJVYSANRY-UHFFFAOYSA-N |  | ||
| CL (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | D [auth A] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| CSD Query on CSD | A | L-PEPTIDE LINKING | C3 H7 N O4 S |  | CYS |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 106.18 | α = 90 |
| b = 106.18 | β = 90 |
| c = 82.88 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| d*TREK | data reduction |
| d*TREK | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Research Foundation in South Africa | South Africa | -- |