Structure of cyclopropane fatty acid synthase from Aquifex aeolicus
Cronan, J.E., Nair, S.K., Lukk, T.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cyclopropane-fatty-acyl-phospholipid synthase | 400 | Aquifex aeolicus VF5 | Mutation(s): 3 Gene Names: cfa, aq_1737 | 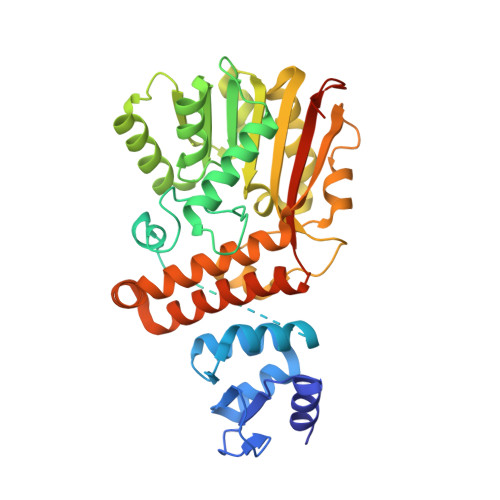 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O67624 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| E8Q (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth A], F [auth B] | 2-azanylethyl-[(2~{S})-2,3-di(hexadecanoyloxy)propoxy]phosphinic acid C37 H74 N O7 P BZYHDQIJHNPDON-DHUJRADRSA-N |  | ||
| SAM (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | G [auth B] | S-ADENOSYLMETHIONINE C15 H22 N6 O5 S MEFKEPWMEQBLKI-FCKMPRQPSA-N |  | ||
| SAH (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | E [auth A] | S-ADENOSYL-L-HOMOCYSTEINE C14 H20 N6 O5 S ZJUKTBDSGOFHSH-WFMPWKQPSA-N |  | ||
| CO3 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | D [auth A], H [auth B] | CARBONATE ION C O3 BVKZGUZCCUSVTD-UHFFFAOYSA-L |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 67.74 | α = 90 |
| b = 77.28 | β = 96.62 |
| c = 86.36 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XSCALE | data scaling |
| PHASER | phasing |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute Of Allergy and Infectious Diseases (NIH/NIAID) | United States | AI15650 |