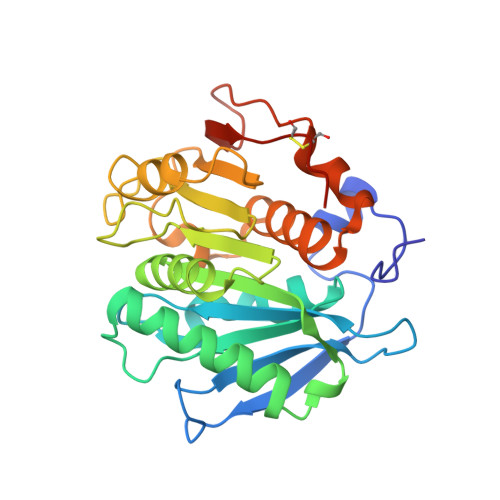
Sourcing thermotolerant poly(ethylene terephthalate) hydrolase scaffolds from natural diversity
Erickson, E., Gado, J.E., Avilan, L., Bratti, F., Brizendine, R.K., Cox, P.A., Gill, R., Graham, R., Kim, D.J., Konig, G., Michener, W.E., Poudel, S., Ramirez, K.J., Shakespeare, T.J., Zahn, M., Boyd, E.S., Payne, C.M., DuBois, J.L., Pickford, A.R., Beckham, G.T., McGeehan, J.E.(2022) Nat Commun 13: 7850