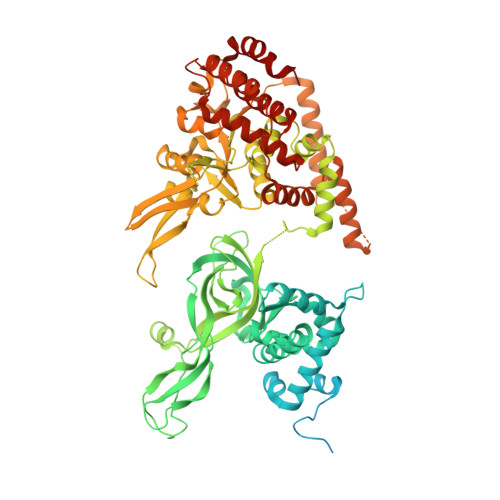
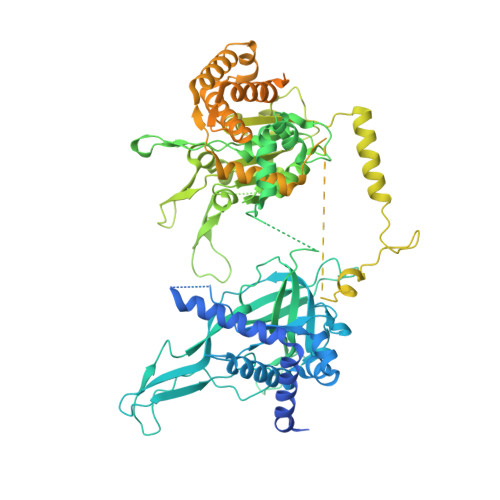
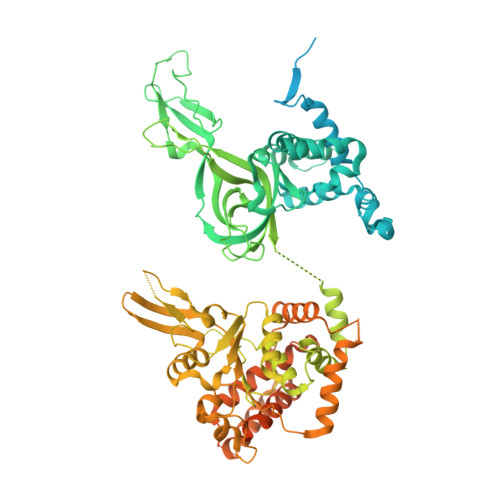
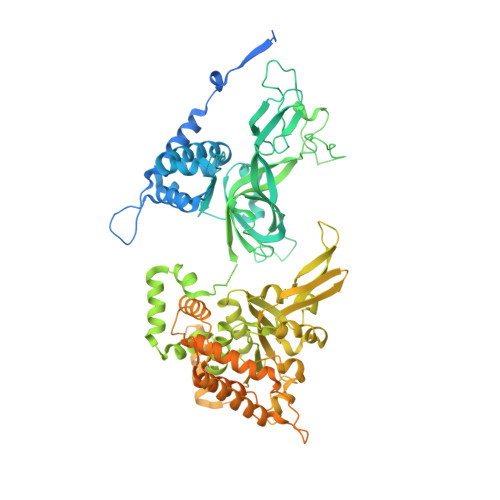
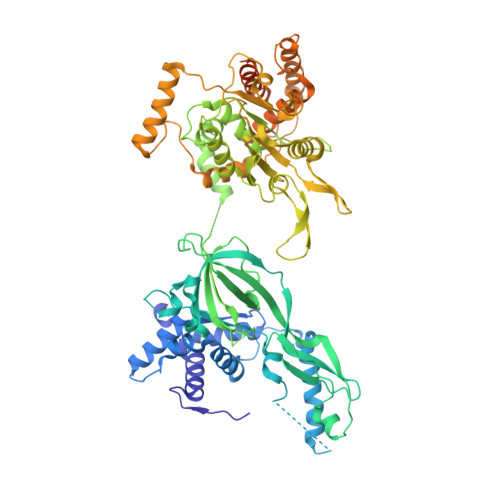
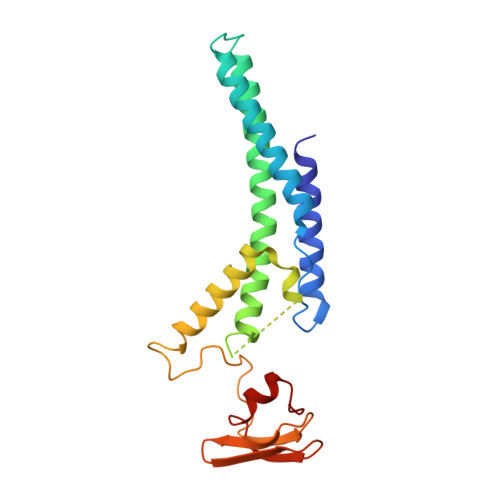
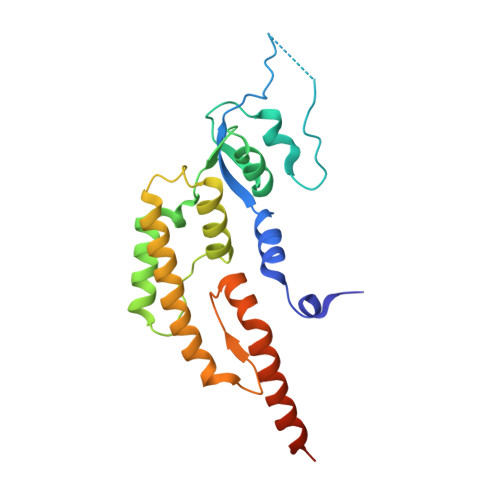
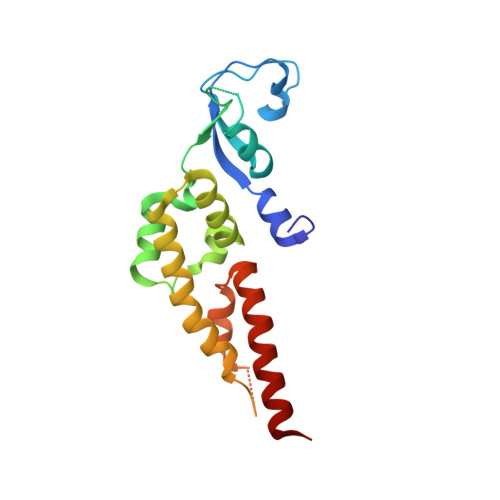
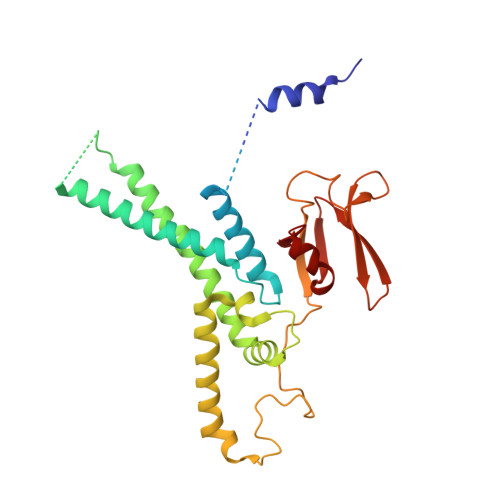
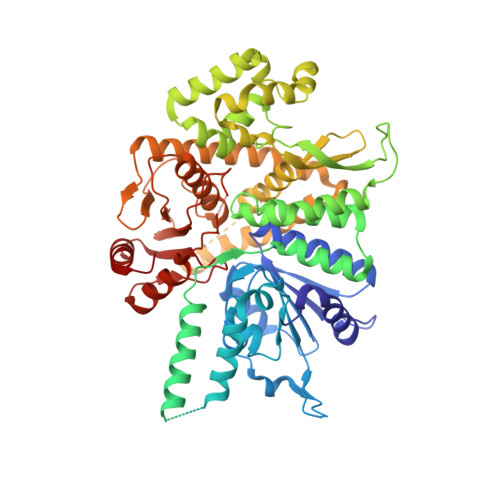
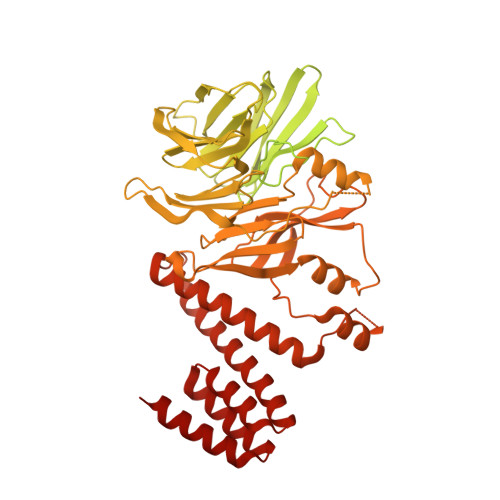
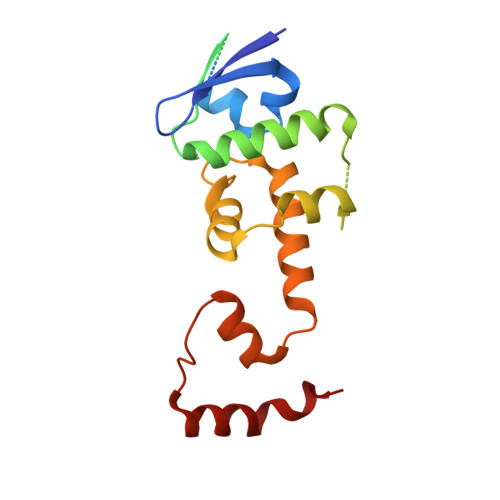
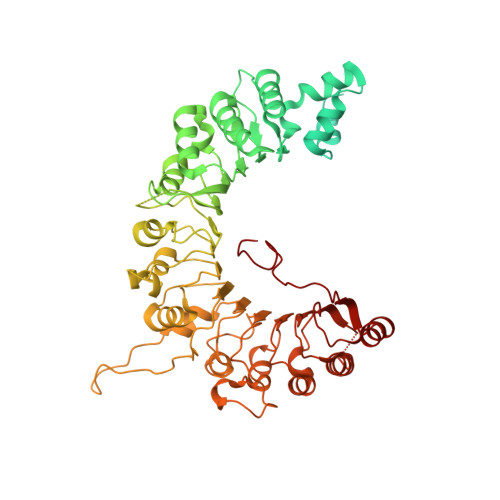
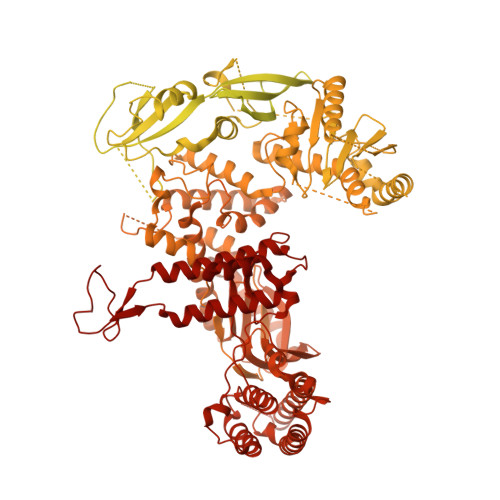
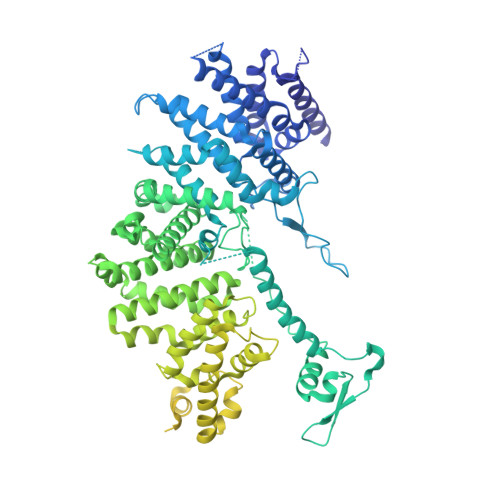
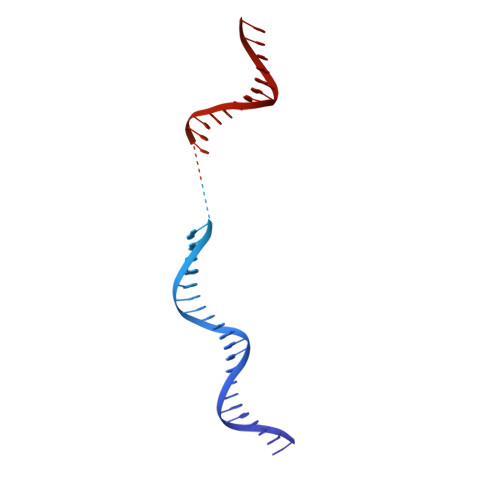
A conserved mechanism for regulating replisome disassembly in eukaryotes.
Jenkyn-Bedford, M., Jones, M.L., Baris, Y., Labib, K.P.M., Cannone, G., Yeeles, J.T.P., Deegan, T.D.(2021) Nature 600: 743-747
- PubMed: 34700328 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-021-04145-3
- Primary Citation Related Structures:
7PLO, 7PMK, 7PMN - PubMed Abstract:
Replisome disassembly is the final step of eukaryotic DNA replication and is triggered by ubiquitylation of the CDC45-MCM-GINS (CMG) replicative helicase 1-3 . Despite being driven by evolutionarily diverse E3 ubiquitin ligases in different eukaryotes (SCF Dia2 in budding yeast 1 , CUL2 LRR1 in metazoa 4-7 ), replisome disassembly is governed by a common regulatory principle, in which ubiquitylation of CMG is suppressed before replication termination, to prevent replication fork collapse. Recent evidence suggests that this suppression is mediated by replication fork DNA 8-10 . However, it is unknown how SCF Dia2 and CUL2 LRR1 discriminate terminated from elongating replisomes, to selectively ubiquitylate CMG only after termination. Here we used cryo-electron microscopy to solve high-resolution structures of budding yeast and human replisome-E3 ligase assemblies. Our structures show that the leucine-rich repeat domains of Dia2 and LRR1 are structurally distinct, but bind to a common site on CMG, including the MCM3 and MCM5 zinc-finger domains. The LRR-MCM interaction is essential for replisome disassembly and, crucially, is occluded by the excluded DNA strand at replication forks, establishing the structural basis for the suppression of CMG ubiquitylation before termination. Our results elucidate a conserved mechanism for the regulation of replisome disassembly in eukaryotes, and reveal a previously unanticipated role for DNA in preserving replisome integrity.
- MRC Laboratory of Molecular Biology, Cambridge, UK.
Organizational Affiliation: