Characterization of Thiamine Diphosphate-Dependent 4-Hydroxybenzoylformate Decarboxylase Enzymes from
Wei, Z., Wilkinson, R.C., Rashid, G.M.M., Brown, D., Fulop, V., Bugg, T.D.H.(2019) Biochemistry 58: 5281-5293
Experimental Data Snapshot
Starting Model: experimental
View more details
(2019) Biochemistry 58: 5281-5293
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Probable benzoylformate decarboxylase | A [auth AAA], B [auth BBB], C [auth CCC], D [auth DDD] | 531 | Rhodococcus jostii RHA1 | Mutation(s): 0 Gene Names: RHA1_ro02985 EC: 4.1.1.7 (PDB Primary Data), 2.2.1.6 (UniProt) | 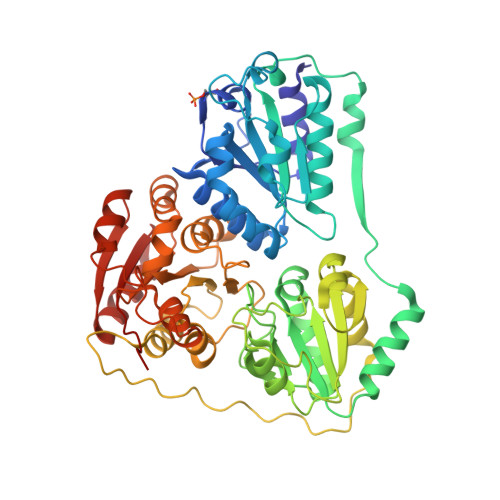 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q0SCE8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| TPP (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | E [auth AAA], G [auth BBB], I [auth CCC], K [auth DDD] | THIAMINE DIPHOSPHATE C12 H19 N4 O7 P2 S AYEKOFBPNLCAJY-UHFFFAOYSA-O |  | ||
| NA Download:Ideal Coordinates CCD File | F [auth AAA], H [auth BBB], J [auth CCC], L [auth DDD] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| SEP Query on SEP | A [auth AAA], B [auth BBB], C [auth CCC], D [auth DDD] | L-PEPTIDE LINKING | C3 H8 N O6 P |  | SER |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 123.5 | α = 90 |
| b = 132.24 | β = 90 |
| c = 138.7 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| XDS | data reduction |
| XDS | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Biotechnology and Biological Sciences Research Council (BBSRC) | United Kingdom | BB/M025772/1 |
| Biotechnology and Biological Sciences Research Council (BBSRC) | United Kingdom | BB/M003523/1 |