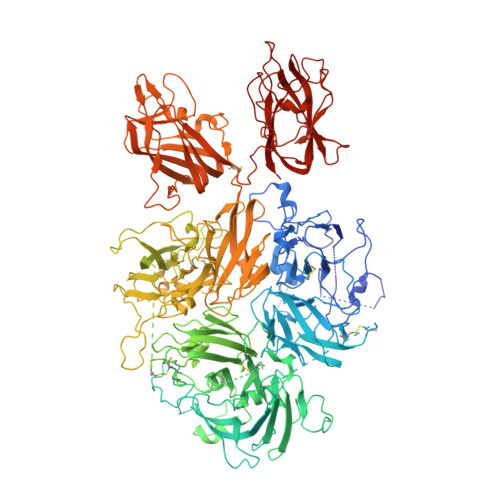
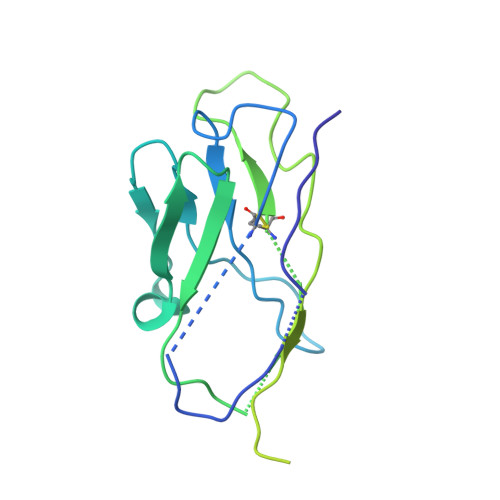
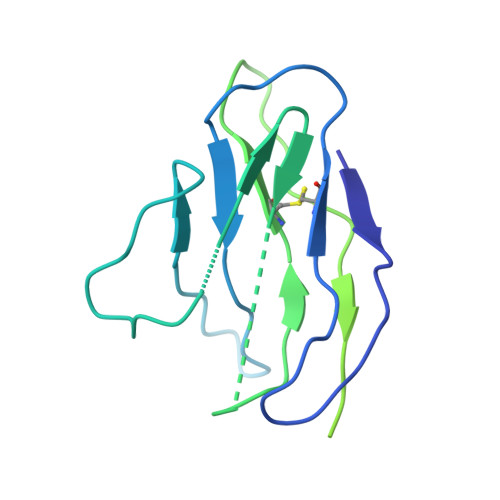
Structure of Blood Coagulation Factor VIII in Complex With an Anti-C2 Domain Non-Classical, Pathogenic Antibody Inhibitor
Ronayne, E.K., Peters, S.C., Gish, J.S., Wilson, C., Spencer, H.T., Doering, C.B., Lollar, P., Spiegel, P.C., Childers, K.C.(2021) Front Immunol 12: 2260