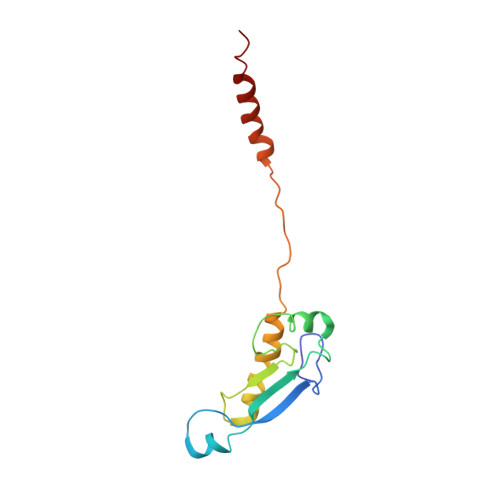
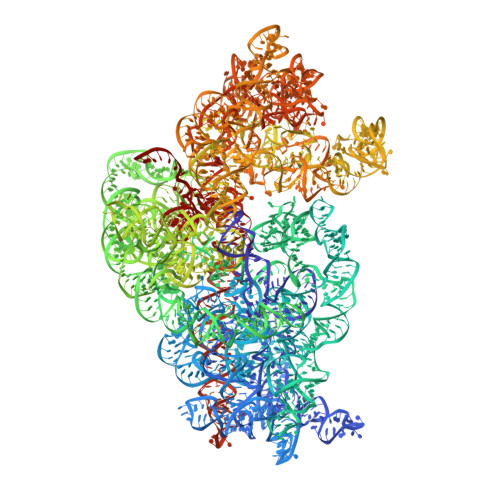
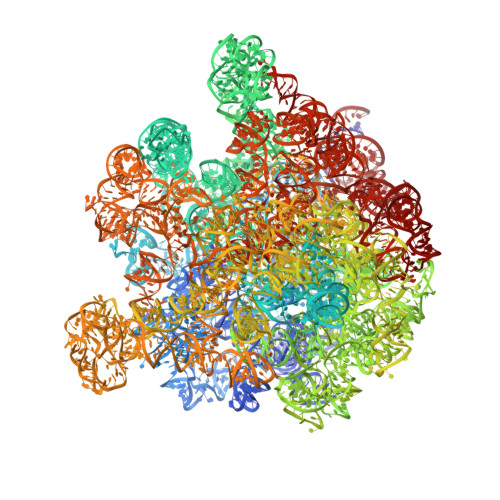
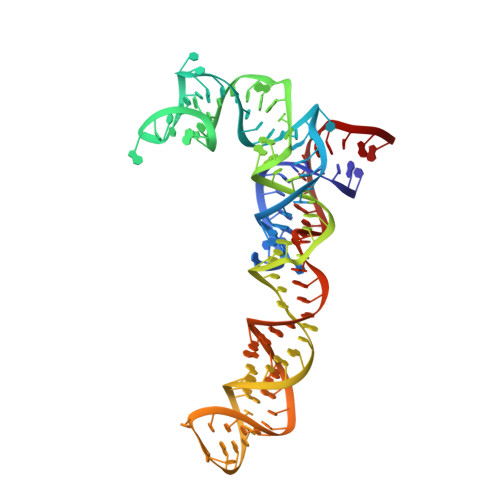
ArfB can displace mRNA to rescue stalled ribosomes.
Carbone, C.E., Demo, G., Madireddy, R., Svidritskiy, E., Korostelev, A.A.(2020) Nat Commun 11: 5552-5552
- PubMed: 33144582 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-020-19370-z
- Primary Citation Related Structures:
7JSS, 7JSW, 7JSZ, 7JT1, 7JT2, 7JT3 - PubMed Abstract:
Ribosomes stalled during translation must be rescued to replenish the pool of translation-competent ribosomal subunits. Bacterial alternative rescue factor B (ArfB) releases nascent peptides from ribosomes stalled on mRNAs truncated at the A site, allowing ribosome recycling. Prior structural work revealed that ArfB recognizes such ribosomes by inserting its C-terminal α-helix into the vacant mRNA tunnel. In this work, we report that ArfB can efficiently recognize a wider range of mRNA substrates, including longer mRNAs that extend beyond the A-site codon. Single-particle cryo-EM unveils that ArfB employs two modes of function depending on the mRNA length. ArfB acts as a monomer to accommodate a shorter mRNA in the ribosomal A site. By contrast, longer mRNAs are displaced from the mRNA tunnel by more than 20 Å and are stabilized in the intersubunit space by dimeric ArfB. Uncovering distinct modes of ArfB function resolves conflicting biochemical and structural studies, and may lead to re-examination of other ribosome rescue pathways, whose functions depend on mRNA lengths.
- RNA Therapeutics Institute, Department of Biochemistry and Molecular Pharmacology, UMass Medical School, Worcester, Massachusetts, 01605, United States.
Organizational Affiliation: