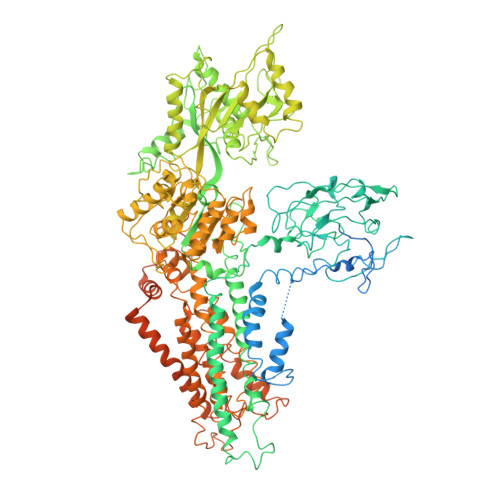
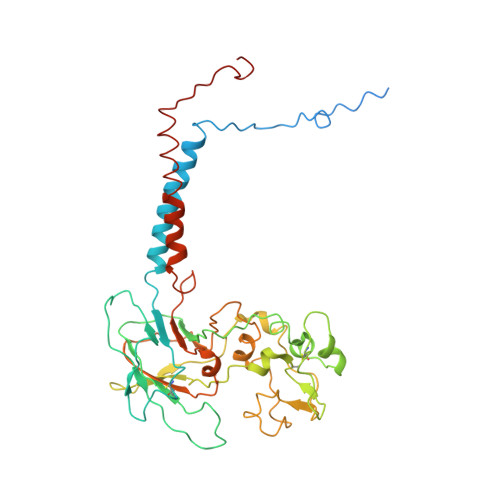
Conformational changes of a phosphatidylcholine flippase in lipid membranes.
Xu, J., He, Y., Wu, X., Li, L.(2022) Cell Rep 38: 110518-110518
- PubMed: 35294892 Search on PubMed
- DOI: https://doi.org/10.1016/j.celrep.2022.110518
- Primary Citation Related Structures:
7DRX, 7DSH, 7DSI, 7F7F, 7WHV, 7WHW - PubMed Abstract:
Type 4 P-type ATPases (P4-ATPases) actively and selectively translocate phospholipids across membrane bilayers. Driven by ATP hydrolysis, P4-ATPases undergo conformational changes during lipid flipping. It is unclear how the active flipping states of P4-ATPases are regulated in the lipid membranes, especially for phosphatidylcholine (PC)-flipping P4-ATPases whose substrate, PC, is a substantial component of membranes. Here, we report the cryoelectron microscopy structures of a yeast PC-flipping P4-ATPase, Dnf1, in lipid environments. In native yeast lipids, Dnf1 adopts a conformation in which the lipid flipping pathway is disrupted. Only when the lipid composition is changed can Dnf1 be captured in the active conformations that enable lipid flipping. These results suggest that, in the native membrane, Dnf1 may stay in an idle conformation that is unable to support the trans-membrane movement of lipids. Dnf1 may have altered conformational preferences in membranes with different lipid compositions.
- State Key Laboratory of Membrane Biology, Peking-Tsinghua Center for Life Sciences, School of Life Sciences, Peking University, Beijing, China.
Organizational Affiliation: