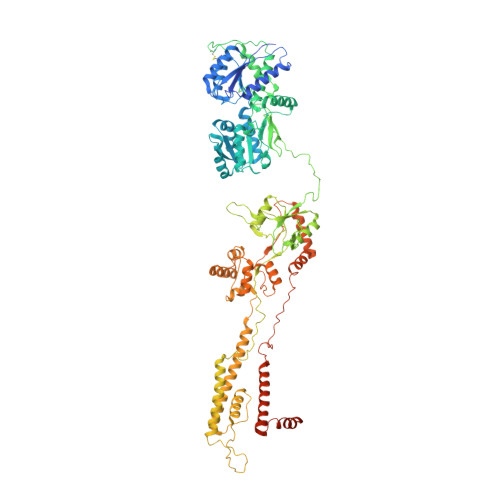
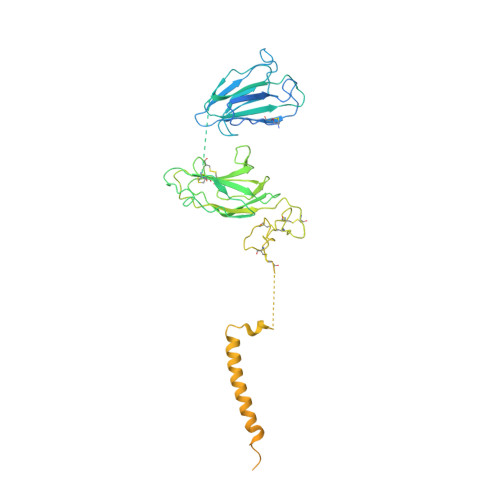
Kainate receptor modulation by NETO2.
He, L., Sun, J., Gao, Y., Li, B., Wang, Y., Dong, Y., An, W., Li, H., Yang, B., Ge, Y., Zhang, X.C., Shi, Y.S., Zhao, Y.(2021) Nature 599: 325-329
- PubMed: 34552241 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-021-03936-y
- Primary Citation Related Structures:
7F56, 7F57, 7F59, 7F5A, 7F5B - PubMed Abstract:
Glutamate-gated kainate receptors are ubiquitous in the central nervous system of vertebrates, mediate synaptic transmission at the postsynapse and modulate transmitter release at the presynapse 1-7 . In the brain, the trafficking, gating kinetics and pharmacology of kainate receptors are tightly regulated by neuropilin and tolloid-like (NETO) proteins 8-11 . Here we report cryo-electron microscopy structures of homotetrameric GluK2 in complex with NETO2 at inhibited and desensitized states, illustrating variable stoichiometry of GluK2-NETO2 complexes, with one or two NETO2 subunits associating with GluK2. We find that NETO2 accesses only two broad faces of kainate receptors, intermolecularly crosslinking the lower lobe of ATD A/C , the upper lobe of LBD B/D and the lower lobe of LBD A/C , illustrating how NETO2 regulates receptor-gating kinetics. The transmembrane helix of NETO2 is positioned proximal to the selectivity filter and competes with the amphiphilic H1 helix after M4 for interaction with an intracellular cap domain formed by the M1-M2 linkers of the receptor, revealing how rectification is regulated by NETO2.
- National Laboratory of Biomacromolecules, CAS Center for Excellence in Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China.
Organizational Affiliation: