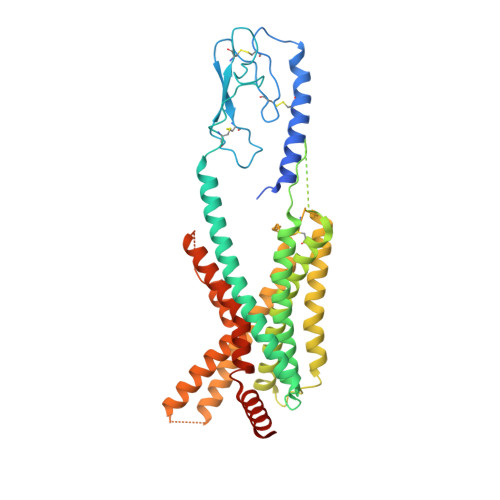
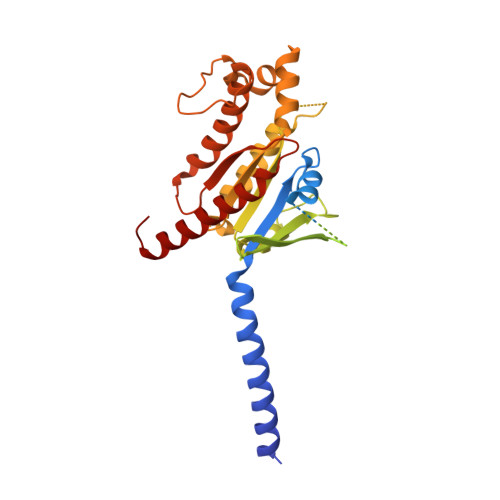
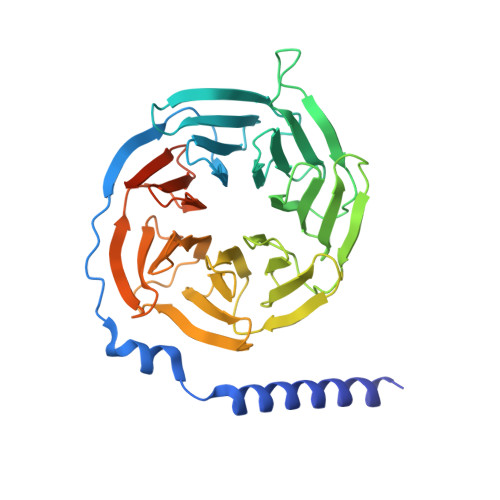
Molecular insights into differentiated ligand recognition of the human parathyroid hormone receptor 2.
Wang, X., Cheng, X., Zhao, L., Wang, Y., Ye, C., Zou, X., Dai, A., Cong, Z., Chen, J., Zhou, Q., Xia, T., Jiang, H., Xu, H.E., Yang, D., Wang, M.W.(2021) Proc Natl Acad Sci U S A 118
- PubMed: 34353904 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.2101279118
- Primary Citation Related Structures:
7F16 - PubMed Abstract:
The parathyroid hormone receptor 2 (PTH2R) is a class B1 G protein-coupled receptor (GPCR) involved in the regulation of calcium transport, nociception mediation, and wound healing. Naturally occurring mutations in PTH2R were reported to cause hereditary diseases, including syndromic short stature. Here, we report the cryogenic electron microscopy structure of PTH2R bound to its endogenous ligand, tuberoinfundibular peptide (TIP39), and a heterotrimeric G s protein at a global resolution of 2.8 Å. The structure reveals that TIP39 adopts a unique loop conformation at the N terminus and deeply inserts into the orthosteric ligand-binding pocket in the transmembrane domain. Molecular dynamics simulation and site-directed mutagenesis studies uncover the basis of ligand specificity relative to three PTH2R agonists, TIP39, PTH, and PTH-related peptide. We also compare the action of TIP39 with an antagonist lacking six residues from the peptide N terminus, TIP(7-39), which underscores the indispensable role of the N terminus of TIP39 in PTH2R activation. Additionally, we unveil that a disease-associated mutation G258D significantly diminished cAMP accumulation induced by TIP39. Together, these results not only provide structural insights into ligand specificity and receptor activation of class B1 GPCRs but also offer a foundation to systematically rationalize the available pharmacological data to develop therapies for various disorders associated with PTH2R.
- The National Center for Drug Screening, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai 201203, China.
Organizational Affiliation: