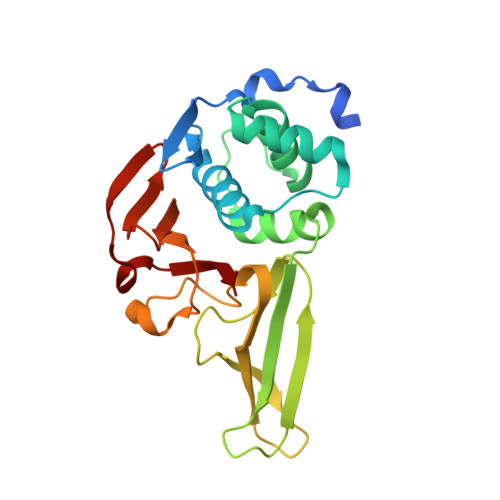
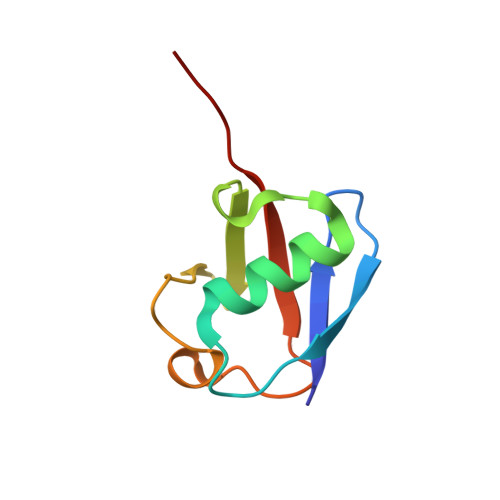
Structural and Biochemical Characterization of Porcine Epidemic Diarrhea Virus Papain-Like Protease 2.
Chu, H.F., Cheng, S.C., Sun, C.Y., Chou, C.Y., Lin, T.H., Chen, W.Y.(2022) J Virol 96: e0137221-e0137221
- PubMed: 34643430 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/JVI.01372-21
- Primary Citation Related Structures:
7F0U - PubMed Abstract:
Coronaviral papain-like proteases (PLpros) are essential enzymes that mediate not only the proteolytic processes of viral polyproteins during virus replication but also the deubiquitination and deISGylation of cellular proteins that attenuate host innate immune responses. Therefore, PLpros are attractive targets for antiviral drug development. Here, we report the crystal structure of papain-like protease 2 (PLP2) of porcine epidemic diarrhea virus (PEDV) in complex with ubiquitin (Ub). The X-ray structural analyses reveal that PEDV PLP2 interacts with the Ub substrate mainly through the Ub core region and C-terminal tail. Mutations of Ub-interacting residues resulted in a moderately or completely abolished deubiquitinylating function of PEDV PLP2. In addition, our analyses also indicate that 2-residue-extended blocking loop 2 at the S4 subsite contributes to the substrate selectivity and binding affinity of PEDV PLP2. Furthermore, the PEDV PLP2 Glu99 residue, conserved in alphacoronavirus PLpros, was found to govern the preference of a positively charged P4 residue of peptidyl substrates. Collectively, our data provided structure-based information for the substrate binding and selectivity of PEDV PLP2. These findings may help us gain insights into the deubiquitinating (DUB) and proteolytic functions of PEDV PLP2 from a structural perspective. IMPORTANCE Current challenges in coronaviruses (CoVs) include a comprehensive understanding of the mechanistic effects of associated enzymes, including the 3C-like and papain-like proteases. We have previously reported that the PEDV PLP2 exhibits a broader substrate preference, superior DUB function, and inferior peptidase activity. However, the structural basis for these functions remains largely unclear. Here, we show the high-resolution X-ray crystal structure of PEDV PLP2 in complex with Ub. Integrated structural and biochemical analyses revealed that (i) three Ub core-interacting residues are essential for DUB function, (ii) 2-residue-elongated blocking loop 2 regulates substrate selectivity, and (iii) a conserved glutamate residue governs the substrate specificity of PEDV PLP2. Collectively, our findings provide not only structural insights into the catalytic mechanism of PEDV PLP2 but also a model for developing antiviral strategies.
- Biomedical Industry Ph.D. Program, National Yang Ming Chiao Tung University, Taipei, Taiwan.
Organizational Affiliation: