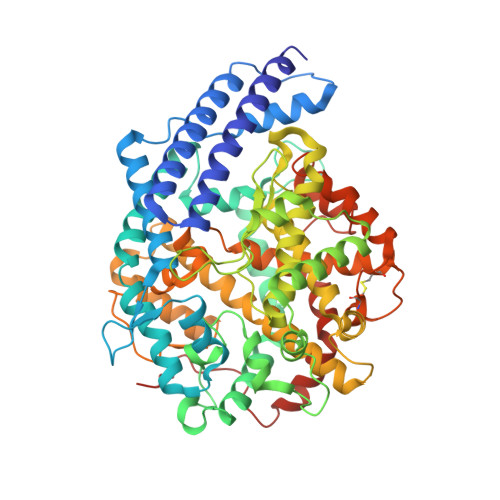
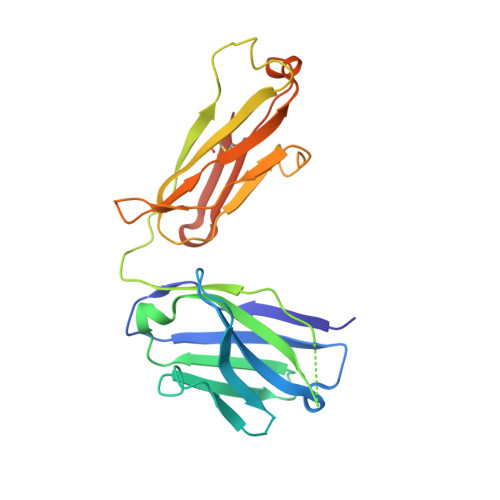
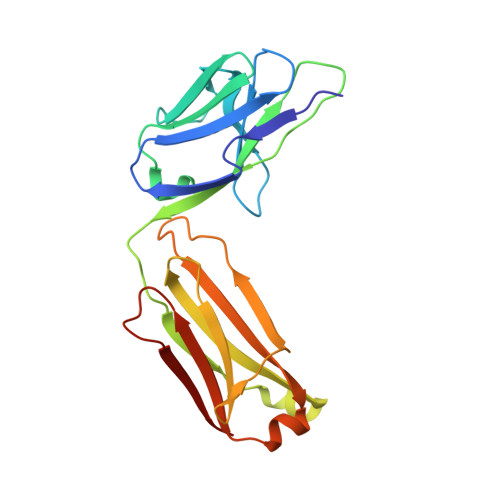
A broadly neutralizing humanized ACE2-targeting antibody against SARS-CoV-2 variants.
Du, Y., Shi, R., Zhang, Y., Duan, X., Li, L., Zhang, J., Wang, F., Zhang, R., Shen, H., Wang, Y., Wu, Z., Peng, Q., Pan, T., Sun, W., Huang, W., Feng, Y., Feng, H., Xiao, J., Tan, W., Wang, Y., Wang, C., Yan, J.(2021) Nat Commun 12: 5000-5000
- PubMed: 34404805
- DOI: https://doi.org/10.1038/s41467-021-25331-x
- Primary Citation Related Structures:
7E7E - PubMed Abstract:
The successive emergences and accelerating spread of novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) lineages and evolved resistance to some ongoing clinical therapeutics increase the risks associated with the coronavirus disease 2019 (COVID-19) pandemic. An urgent intervention for broadly effective therapies to limit the morbidity and mortality of COVID-19 and future transmission events from SARS-related coronaviruses (SARSr-CoVs) is needed. Here, we isolate and humanize an angiotensin-converting enzyme-2 (ACE2)-blocking monoclonal antibody (MAb), named h11B11, which exhibits potent inhibitory activity against SARS-CoV and circulating global SARS-CoV-2 lineages. When administered therapeutically or prophylactically in the hACE2 mouse model, h11B11 alleviates and prevents SARS-CoV-2 replication and virus-induced pathological syndromes. No significant changes in blood pressure and hematology chemistry toxicology were observed after injections of multiple high dosages of h11B11 in cynomolgus monkeys. Analysis of the structures of the h11B11/ACE2 and receptor-binding domain (RBD)/ACE2 complexes shows hindrance and epitope competition of the MAb and RBD for the receptor. Together, these results suggest h11B11 as a potential therapeutic countermeasure against SARS-CoV, SARS-CoV-2, and escape variants.
- Key Laboratory of Molecular Biophysics of the Ministry of Education, National Engineering Research Center for Nanomedicine, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan, China.
Organizational Affiliation: