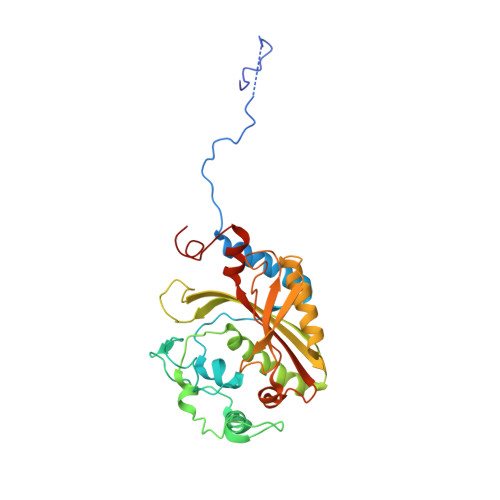
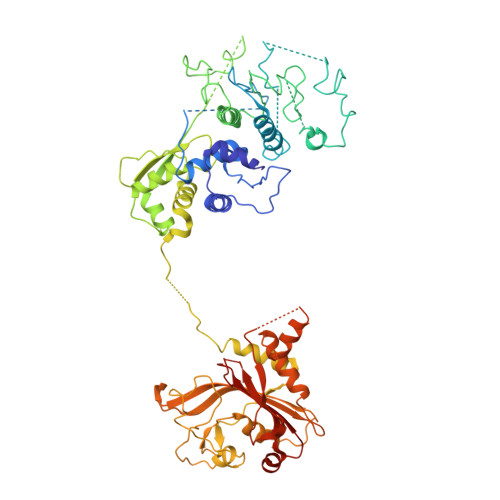
Inhibition of Plasmodium falciparum phenylalanine tRNA synthetase provides opportunity for antimalarial drug development.
Sharma, M., Mutharasappan, N., Manickam, Y., Harlos, K., Melillo, B., Comer, E., Tabassum, H., Parvez, S., Schreiber, S.L., Sharma, A.(2022) Structure 30: 962-972.e3
- PubMed: 35460612
- DOI: https://doi.org/10.1016/j.str.2022.03.017
- Primary Citation Related Structures:
7DPI - PubMed Abstract:
Bicyclic azetidine compounds possess antimalarial activity via targeting of the cytoplasmic Plasmodium falciparum (Pf) protein translation enzyme phenylalanine-tRNA synthetase (cFRS). These drugs kill parasites both in vitro and in vivo, including the blood, liver, and transmission developmental stages. Here we present the co-crystal structure of PfcFRS with a potent inhibitor, the bicyclic azetidine BRD7929. Our studies reveal high-affinity binding of BRD7929 with PfcFRS along with exquisite specificity compared with the human enzyme, leading in turn to potent and selective inhibition of the parasite enzyme. Our co-crystal structure shows that BRD7929 binds in the active site in the α subunit of PfcFRS, where it occupies the amino acid site, an auxiliary site, and partially the ATP site. This structural snapshot of inhibitor-bound PfcFRS thus provides a platform for the structure-guided optimization of novel antimalarial compounds.
- Molecular Medicine, Structural Parasitology Group, International Centre for Genetic Engineering and Biotechnology (ICGEB), Aruna Asaf Ali Marg, New Delhi 110067, India; Department of Medical Elementology and Toxicology, School of Chemical and Life Sciences, Jamia Hamdard, New Delhi 110062, India.
Organizational Affiliation: