Reprogramming Epoxide Hydrolase to Improve Enantioconvergence in Hydrolysis of Styrene Oxide Scaffolds
Li, F.L., Qiu, Y.Y., Zheng, Y.C., Chen, F.F., Kong, X.D., Xu, J.H., Yu, H.L.(2021) Adv Synth Catal 362: 4699-4706
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
(2021) Adv Synth Catal 362: 4699-4706
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Epoxide hydrolase | A [auth B] | 328 | Vigna radiata | Mutation(s): 1 Gene Names: EH2 EC: 3.3.2.10 | 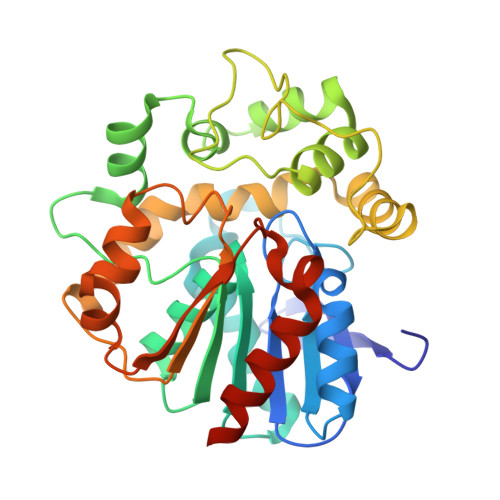 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A0R5NGA4 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 70.813 | α = 90 |
| b = 70.813 | β = 90 |
| c = 127.377 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data scaling |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| HKL-2000 | data reduction |
| PHASER | phasing |
| REFMAC | refinement |