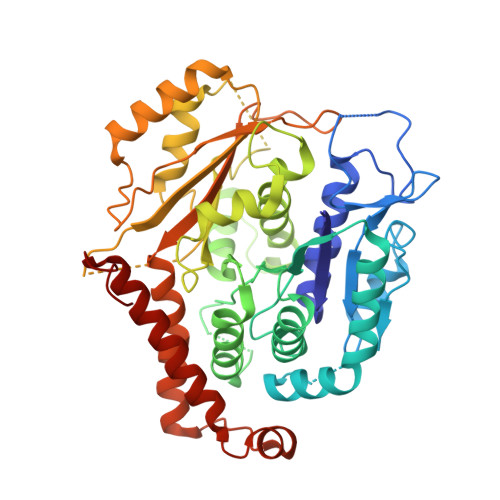
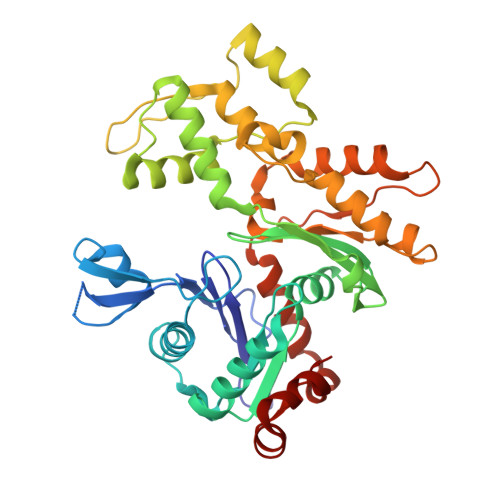
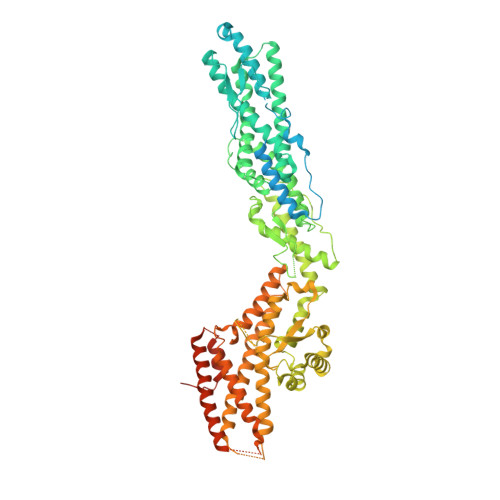
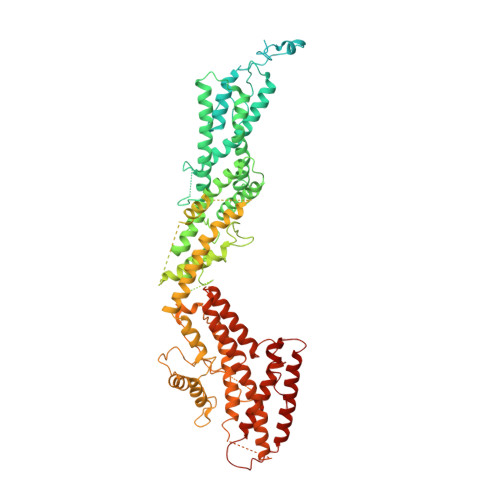
Assembly of the asymmetric human gamma-tubulin ring complex by RUVBL1-RUVBL2 AAA ATPase.
Zimmermann, F., Serna, M., Ezquerra, A., Fernandez-Leiro, R., Llorca, O., Luders, J.(2020) Sci Adv 6
- PubMed: 33355144 Search on PubMed
- DOI: https://doi.org/10.1126/sciadv.abe0894
- Primary Citation Related Structures:
7AS4 - PubMed Abstract:
The microtubule nucleator γ-tubulin ring complex (γTuRC) is essential for the function of microtubule organizing centers such as the centrosome. Since its discovery over two decades ago, γTuRC has evaded in vitro reconstitution and thus detailed structure-function studies. Here, we show that a complex of RuvB-like protein 1 (RUVBL1) and RUVBL2 "RUVBL" controls assembly and composition of γTuRC in human cells. Likewise, RUVBL assembles γTuRC from a minimal set of core subunits in a heterologous coexpression system. RUVBL interacts with γTuRC subcomplexes but is not part of fully assembled γTuRC. Purified, reconstituted γTuRC has nucleation activity and resembles native γTuRC as revealed by its cryo-electron microscopy (cryo-EM) structure at ~4.0-Å resolution. We further use cryo-EM to identify features that determine the intricate, higher-order γTuRC architecture. Our work finds RUVBL as an assembly factor that regulates γTuRC in cells and allows production of recombinant γTuRC for future in-depth mechanistic studies.
- Mechanisms of Disease Programme, Institute for Research in Biomedicine (IRB Barcelona), The Barcelona Institute of Science and Technology (BIST), Baldiri Reixac 10, 08028 Barcelona, Spain.
Organizational Affiliation: