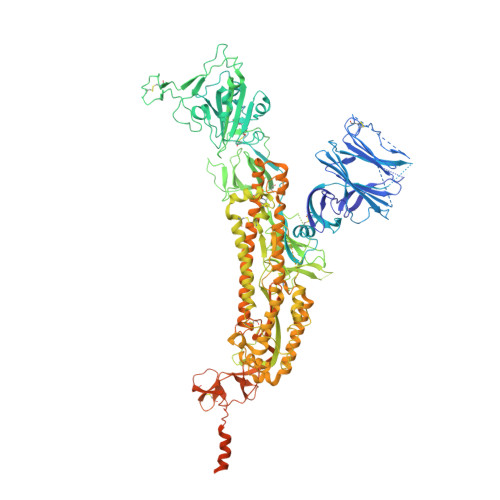
Structure-Based Design with Tag-Based Purification and In-Process Biotinylation Enable Streamlined Development of SARS-CoV-2 Spike Molecular Probes.
Zhou, T., Teng, I.T., Olia, A.S., Cerutti, G., Gorman, J., Nazzari, A., Shi, W., Tsybovsky, Y., Wang, L., Wang, S., Zhang, B., Zhang, Y., Katsamba, P.S., Petrova, Y., Banach, B.B., Fahad, A.S., Liu, L., Acevedo, S.N.L., Madan, B., de Souza, M.O., Pan, X., Wang, P., Wolfe, J.R., Yin, M., Ho, D.D., Phung, E., DiPiazza, A., Chang, L., Abiona, O., Corbett, K.S., DeKosky, B.J., Graham, B.S., Mascola, J.R., Misasi, J., Ruckwardt, T., Sullivan, N.J., Shapiro, L., Kwong, P.D.(2020) SSRN : 3639618-3639618
- PubMed: 32742241 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.2139/ssrn.3639618
- Primary Citation Related Structures:
6XF5, 6XF6 - PubMed Abstract:
Biotin-labeled molecular probes, comprising specific regions of the SARS-CoV-2 spike, would be helpful in the isolation and characterization of antibodies targeting this recently emerged pathogen. To develop such probes, we designed constructs incorporating an N-terminal purification tag, a site-specific protease-cleavage site, the probe region of interest, and a C-terminal sequence targeted by biotin ligase. Probe regions included full-length spike ectodomain as well as various subregions, and we also designed mutants to eliminate recognition of the ACE2 receptor. Yields of biotin-labeled probes from transient transfection ranged from ~0.5 mg/L for the complete ectodomain to >5 mg/L for several subregions. Probes were characterized for antigenicity and ACE2 recognition, and the structure of the spike ectodomain probe was determined by cryo-electron microscopy. We also characterized antibody-binding specificities and cell-sorting capabilities of the biotinylated probes. Altogether, structure-based design coupled to efficient purification and biotinylation processes can thus enable streamlined development of SARS-CoV-2 spike-ectodomain probes. Funding: Support for this work was provided by the Intramural Research Program of the Vaccine Research Center, National Institute of Allergy and Infectious Diseases (NIAID). Support for this work was also provided by COVID-19 Fast Grants, the Jack Ma Foundation, the Self Graduate Fellowship Program, and NIH grants DP5OD023118, R21AI143407, and R21AI144408. Some of this work was performed at the Columbia University Cryo-EM Center at the Zuckerman Institute, and some at the Simons Electron Microscopy Center (SEMC) and National Center for Cryo-EM Access and Training (NCCAT) located at the New York Structural Biology Center, supported by grants from the Simons Foundation (SF349247), NYSTAR, and the NIH National Institute of General Medical Sciences (GM103310). Conflict of Interest: The authors declare that they have no conflict of interest. Ethical Approval: Peripheral blood mononuclear cells (PBMCs) for B cell sorting were obtained from a convalescent SARS-CoV-2 patient (collected 75 days post symptom onset under an IRB approved clinical trial protocol, VRC 200 - ClinicalTrials.gov Identifier: NCT00067054) and a healthy control donor from the NIH blood bank pre-SARS-CoV-2 pandemic.
- Vaccine Research Center, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD 20892, USA.
Organizational Affiliation: