DNMT1 activity, base flipping mechanism and genome-wide DNA methylation are regulated by the DNA sequence context
Adam, S., Anteneh, H., Hornisch, M., Wagner, V., Lu, J., Radde, N.E., Bashtrykov, P., Song, J., Jeltsch, A.(2020) Nat Commun
Experimental Data Snapshot
Starting Model: experimental
View more details
(2020) Nat Commun
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA (cytosine-5)-methyltransferase 1 | 873 | Mus musculus | Mutation(s): 0 Gene Names: Dnmt1, Dnmt, Met1, Uim EC: 2.1.1.37 | 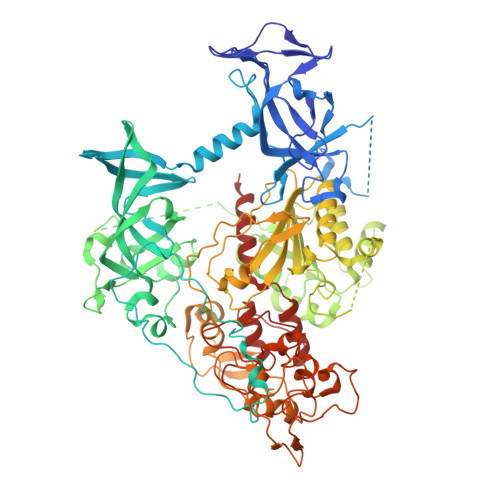 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P13864 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| CCG DNA (5'-D(*AP*CP*TP*TP*AP*(5CM)P*GP*GP*AP*AP*GP*G)-3') | C, D [auth E] | 12 | synthetic construct | 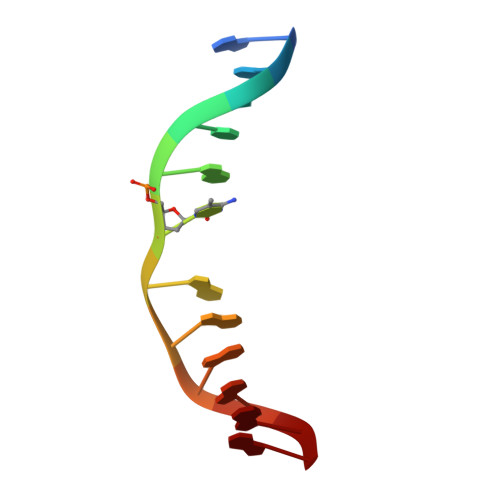 |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
Entity ID: 3 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| CCG DNA (5'-D(*CP*CP*TP*TP*CP*(C49)P*GP*TP*AP*AP*GP*T)-3') | E [auth D], F | 12 | synthetic construct | 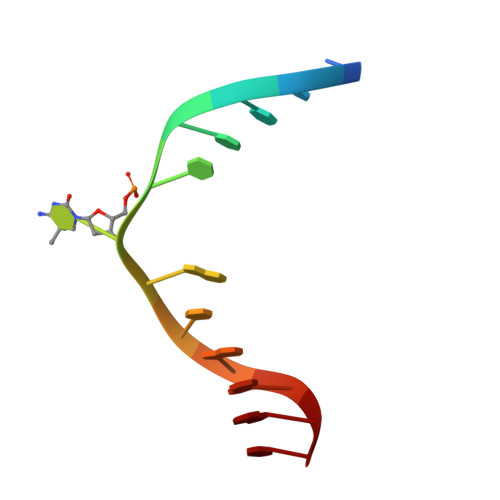 |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SAH (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | G [auth A], J [auth B] | S-ADENOSYL-L-HOMOCYSTEINE C14 H20 N6 O5 S ZJUKTBDSGOFHSH-WFMPWKQPSA-N |  | ||
| ZN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | H [auth A], I [auth A], K [auth B], L [auth B] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 89.794 | α = 90 |
| b = 152.593 | β = 94.74 |
| c = 95.631 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | 1R35GM119721 |