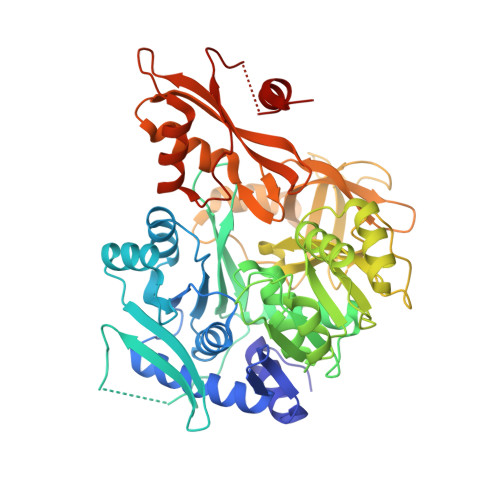
The identification and characterization of an oxalyl-CoA synthetase from grass pea ( Lathyrus sativus L.).
Goldsmith, M., Barad, S., Peleg, Y., Albeck, S., Dym, O., Brandis, A., Mehlman, T., Reich, Z.(2022) RSC Chem Biol 3: 320-333
- PubMed: 35359497 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1039/d1cb00202c
- Primary Citation Related Structures:
6QJZ - PubMed Abstract:
Oxalic acid is a small metabolite found in many plants. It serves as protection from herbivores, a chelator of metal ions, a regulator of calcium levels, and additional tasks. However, it is also a strong di-carboxylic acid that can compromise plant viability by reducing cellular pH. Several metabolic pathways have evolved to control oxalate levels in plants by enzymatic degradation. Among them is the pathway that utilizes oxalyl-CoA synthetase (OCS, EC 6.2.1.8) and ATP to convert oxalate to oxalyl-CoA. Oxalyl-CoA can then be degraded to CO 2 or utilized as a precursor for the synthesis of other compounds. In grass pea ( Lathyrus sativus L.), a grain legume grown in Asia and Africa for human and animal consumption, the neurotoxic compound β- N -oxalyl-l-α,β-diaminopropionic acid (β-ODAP) is synthesized from oxalyl-CoA and l-α,β-diaminopropionic acid (l-DAPA). Here, we report on the identification and characterization of oxalyl CoA-synthetase from grass pea ( Ls OCS). The gene encoding Ls OCS was amplified from grass pea, and then expressed and purified from E. coli cells as an untagged, monomeric protein of 56 kDa. Its catalytic efficiency with oxalate, K oxalate M = 71.5 ± 13.3 μM, V max = 8.2 ± 0.8 μmole min -1 mg -1 , was similar to that of OCS homologs from Arabidopsis thaliana ( At AAE3) and Medicago truncatula ( Mt AAE3). The enzyme was crystalized in complex with AMP and is the first OCS whose structure was determined in the thioester-forming conformation. Finally, we propose that substituting Ls OCS with an oxalate oxidase or decarboxylase may reduce the levels of β-ODAP in grass pea.
- Dept. of Biomolecular Sciences, Weizmann Institute of Science Rehovot 7610001 Israel moshe.goldsmith@weizmann.ac.il ziv.reich@weizmann.ac.il +972-8-9344118 +972-8-9343278 +972-8-9342982.
Organizational Affiliation: