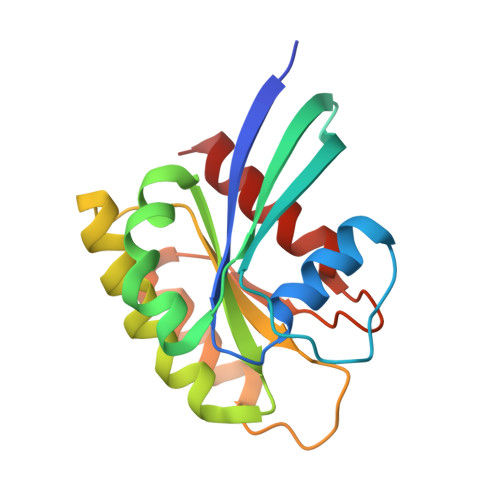
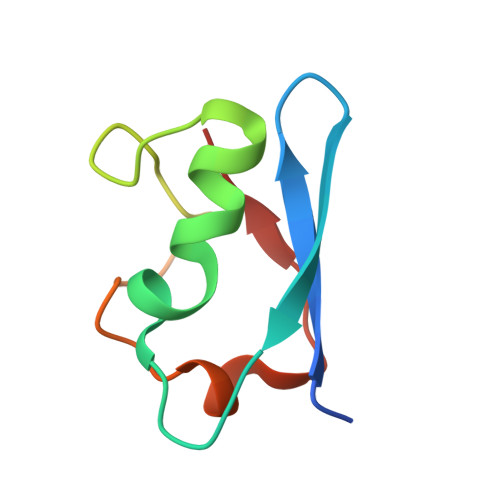
Conformation-specific inhibitors of activated Ras GTPases reveal limited Ras dependency of patient-derived cancer organoids.
Wiechmann, S., Maisonneuve, P., Grebbin, B.M., Hoffmeister, M., Kaulich, M., Clevers, H., Rajalingam, K., Kurinov, I., Farin, H.F., Sicheri, F., Ernst, A.(2020) J Biological Chem 295: 4526-4540
- PubMed: 32086379 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.RA119.011025
- Primary Citation Related Structures:
6NTC, 6NTD - PubMed Abstract:
The small GTPases H, K, and NRAS are molecular switches indispensable for proper regulation of cellular proliferation and growth. Several mutations in the genes encoding members of this protein family are associated with cancer and result in aberrant activation of signaling processes caused by a deregulated recruitment of downstream effector proteins. In this study, we engineered variants of the Ras-binding domain (RBD) of the C-Raf proto-oncogene, Ser/Thr kinase (CRAF). These variants bound with high affinity with the effector-binding site of Ras in an active conformation. Structural characterization disclosed how the newly identified RBD mutations cooperate and thereby enhance affinity with the effector-binding site in Ras compared with WT RBD. The engineered RBD variants closely mimicked the interaction mode of naturally occurring Ras effectors and acted as dominant-negative affinity reagents that block Ras signal transduction. Experiments with cancer cells showed that expression of these RBD variants inhibits Ras signaling, reducing cell growth and inducing apoptosis. Using these optimized RBD variants, we stratified patient-derived colorectal cancer organoids with known Ras mutational status according to their response to Ras inhibition. These results revealed that the presence of Ras mutations was insufficient to predict sensitivity to Ras inhibition, suggesting that not all of these tumors required Ras signaling for proliferation. In summary, by engineering the Ras/Raf interface of the CRAF-RBD, we identified potent and selective inhibitors of Ras in its active conformation that outcompete binding of Ras-signaling effectors.
- Institute of Biochemistry II, Goethe University Frankfurt-Medical Faculty, University Hospital, 60596 Frankfurt am Main, Germany.
Organizational Affiliation: