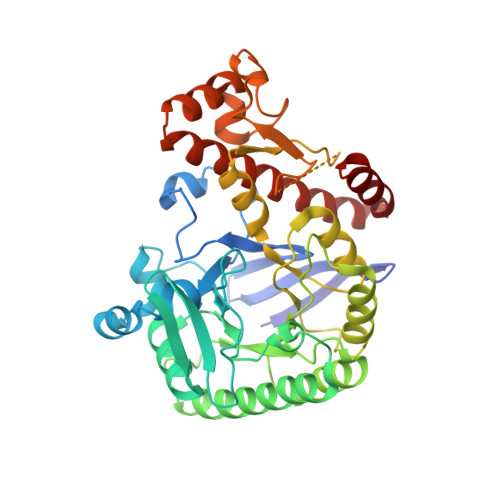
Homodimer Architecture of QTRT2, the Noncatalytic Subunit of the Eukaryotic tRNA-Guanine Transglycosylase.
Behrens, C., Biela, I., Petiot-Becard, S., Botzanowski, T., Cianferani, S., Sager, C.P., Klebe, G., Heine, A., Reuter, K.(2018) Biochemistry 57: 3953-3965
- PubMed: 29862811
- DOI: https://doi.org/10.1021/acs.biochem.8b00294
- Primary Citation of Related Structures:
6FV5 - PubMed Abstract:
The bacterial enzyme tRNA-guanine transglycosylase (TGT) is involved in the biosynthesis of queuosine, a modified nucleoside present in the anticodon wobble position of tRNA His , tRNA Tyr , tRNA Asp , and tRNA Asn . Although it forms a stable homodimer endowed with two active sites, it is, for steric reasons, able to bind and convert only one tRNA molecule at a time. In contrast, its mammalian counterpart constitutes a heterodimer consisting of a catalytic and a noncatalytic subunit, termed QTRT1 and QTRT2, respectively. Both subunits are homologous to the bacterial enzyme, yet only QTRT1 possesses all the residues required for substrate binding and catalysis. In mice, genetic inactivation of the TGT results in the uncontrolled oxidation of tetrahydrobiopterin and, accordingly, phenylketonuria-like symptoms. For this reason and because of the recent finding that mammalian TGT may be utilized for the treatment of multiple sclerosis, this enzyme is of potential medical relevance, rendering detailed knowledge of its biochemistry and structural architecture highly desirable. In this study, we performed the kinetic characterization of the murine enzyme, investigated potential quaternary structures of QTRT1 and QTRT2 via noncovalent mass spectrometry, and, finally, determined the crystal structure of the murine noncatalytic TGT subunit, QTRT2. In the crystal, QTRT2 is clearly present as a homodimer that is strikingly similar to that formed by bacterial TGT. In particular, a cluster of four aromatic residues within the interface of the bacterial TGT, which constitutes a "hot spot" for dimer stability, is present in a similar constellation in QTRT2.
- Institut für Pharmazeutische Chemie , Philipps-Universität Marburg , Marbacher Weg 6 , D-35032 Marburg , Germany.
Organizational Affiliation: