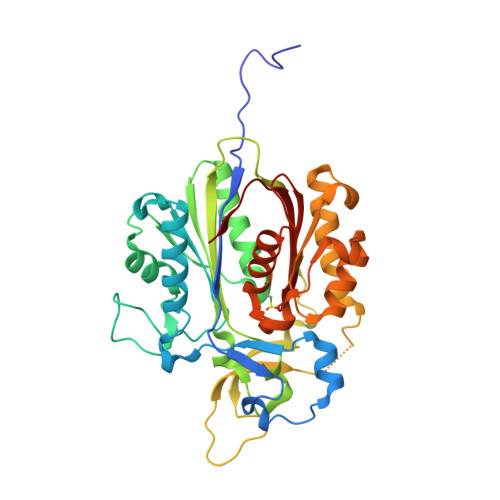
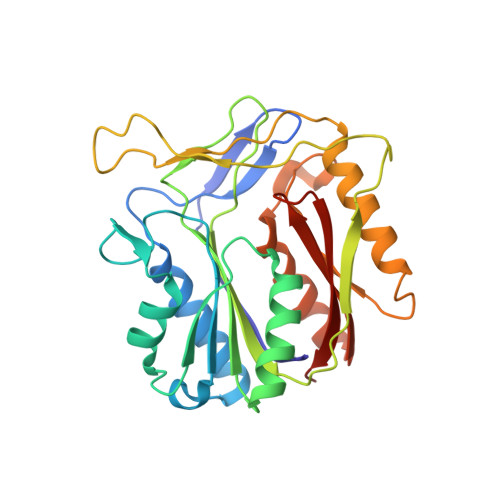
The Alkylquinolone Repertoire of Pseudomonas aeruginosa is Linked to Structural Flexibility of the FabH-like 2-Heptyl-3-hydroxy-4(1H)-quinolone (PQS) Biosynthesis Enzyme PqsBC.
Witzgall, F., Depke, T., Hoffmann, M., Empting, M., Bronstrup, M., Muller, R., Blankenfeldt, W.(2018) Chembiochem 19: 1531-1544
- PubMed: 29722462 Search on PubMed
- DOI: https://doi.org/10.1002/cbic.201800153
- Primary Citation Related Structures:
6ESZ, 6ET0, 6ET1, 6ET2, 6ET3, 6ETO - PubMed Abstract:
Pseudomonas aeruginosa is a bacterial pathogen that causes life-threatening infections in immunocompromised patients. It produces a large armory of saturated and mono-unsaturated 2-alkyl-4(1H)-quinolones (AQs) and AQ N-oxides (AQNOs) that serve as signaling molecules to control the production of virulence factors and that are involved in membrane vesicle formation and iron chelation; furthermore, they also have, for example, antibiotic properties. It has been shown that the β-ketoacyl-acyl-carrier protein synthase III (FabH)-like heterodimeric enzyme PqsBC catalyzes the last step in the biosynthesis of the most abundant AQ congener, 2-heptyl-4(1H)-quinolone (HHQ), by condensing octanoyl-coenzyme A (CoA) with 2-aminobenzoylacetate (2-ABA), but the basis for the large number of other AQs/AQNOs produced by P. aeruginosa is not known. Here, we demonstrate that PqsBC uses different medium-chain acyl-CoAs to produce various saturated AQs/AQNOs and that it also biosynthesizes mono-unsaturated congeners. Further, we determined the structures of PqsBC in four different crystal forms at 1.5 to 2.7 Å resolution. Together with a previous report, the data reveal that PqsBC adopts open, intermediate, and closed conformations that alter the shape of the acyl-binding cavity and explain the promiscuity of PqsBC. The different conformations also allow us to propose a model for structural transitions that accompany the catalytic cycle of PqsBC that might have broader implications for other FabH-enzymes, for which such structural transitions have been postulated but have never been observed.
- Department Structure and Function of Proteins, Helmholtz Centre for Infection Research, Inhoffenstrasse 7, 38124, Braunschweig, Germany.
Organizational Affiliation: