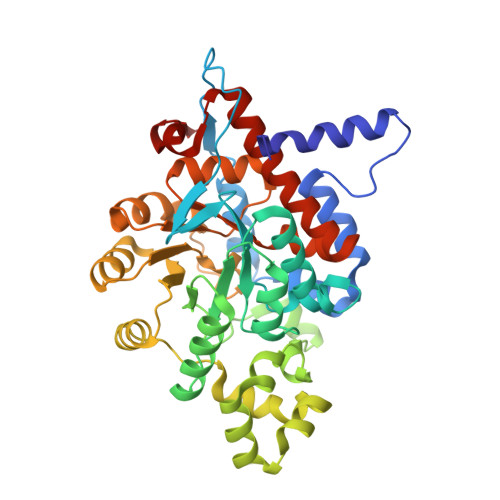
Structure and role for active site lid of lactate monooxygenase from Mycobacterium smegmatis.
Kean, K.M., Karplus, P.A.(2019) Protein Sci 28: 135-149
- PubMed: 30207005 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/pro.3506
- Primary Citation Related Structures:
6DVH, 6DVI - PubMed Abstract:
Lactate monooxygenase (LMO) catalyzes the FMN-dependent "coupled" oxidation of lactate and O 2 to acetate, carbon dioxide, and water, involving pyruvate and hydrogen peroxide as enzyme-bound intermediates. Other α-hydroxy acid oxidase family members follow an "uncoupled pathway," wherein the α-keto acid product quickly dissociates before the reduced flavin reacts with oxygen. Here, we report the structures of Mycobacterium smegmatis wild-type LMO and a wild-type-like C203A variant at 2.1 Å and 1.7 Å resolution, respectively. The overall LMO fold and active site organization, including a bound sulfate mimicking substrate, resemble those of other α-hydroxy acid oxidases. Based on structural similarity, LMO is similarly distant from lactate oxidase, glycolate oxidase, mandelate dehydrogenase, and flavocytochrome b 2 and is the first representative enzyme of its type. Comparisons with other α-hydroxy acid oxidases reveal that LMO has a longer and more compact folded active site loop (Loop 4), which is known in related flavoenzymes to undergo order/disorder transitions to allow substrate/product binding and release. We propose that LMO's Loop 4 has an enhanced stability that is responsible for the slow product release requisite for the coupled pathway. We also note electrostatic features of the LMO active site that promote substrate binding. Whereas the physiological role of LMO remains unknown, we document what can currently be assessed of LMO's distribution in nature, including its unexpected occurrence, presumably through horizontal gene transfer, in halophilic archaea and in a limited group of fungi of the genus Beauveria. BROAD STATEMENT OF IMPACT: This first crystal structure of the FMN-dependent α-hydroxy acid oxidase family member lactate monooxygenase (LMO) reveals it has a uniquely large active site lid that we hypothesize is stable enough to explain the slow dissociation of pyruvate that leads to its "coupled" oxidation of lactate and O 2 to produce acetate, carbon dioxide, and water. Also, the relatively widespread distribution of putative LMOs supports their importance and provides new motivation for their further study.
- Department of Biochemistry and Biophysics, 2011 Agriculture and Life Sciences Building, Oregon State University, Corvallis, Oregon 97331.
Organizational Affiliation: