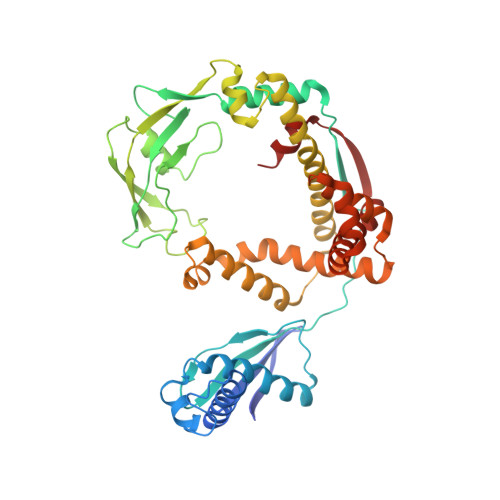
Oligomerization of a molecular chaperone modulates its activity.
Saio, T., Kawagoe, S., Ishimori, K., Kalodimos, C.G.(2018) Elife 7
- PubMed: 29714686 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.35731
- Primary Citation Related Structures:
6D6S - PubMed Abstract:
Molecular chaperones alter the folding properties of cellular proteins via mechanisms that are not well understood. Here, we show that Trigger Factor (TF), an ATP-independent chaperone, exerts strikingly contrasting effects on the folding of non-native proteins as it transitions between a monomeric and a dimeric state. We used NMR spectroscopy to determine the atomic resolution structure of the 100 kDa dimeric TF. The structural data show that some of the substrate-binding sites are buried in the dimeric interface, explaining the lower affinity for protein substrates of the dimeric compared to the monomeric TF. Surprisingly, the dimeric TF associates faster with proteins and it exhibits stronger anti-aggregation and holdase activity than the monomeric TF. The structural data show that the dimer assembles in a way that substrate-binding sites in the two subunits form a large contiguous surface inside a cavity, thus accounting for the observed accelerated association with unfolded proteins. Our results demonstrate how the activity of a chaperone can be modulated to provide distinct functional outcomes in the cell.
- Department of Chemistry, Faculty of Science, Hokkaido University, Sapporo, Japan.
Organizational Affiliation: