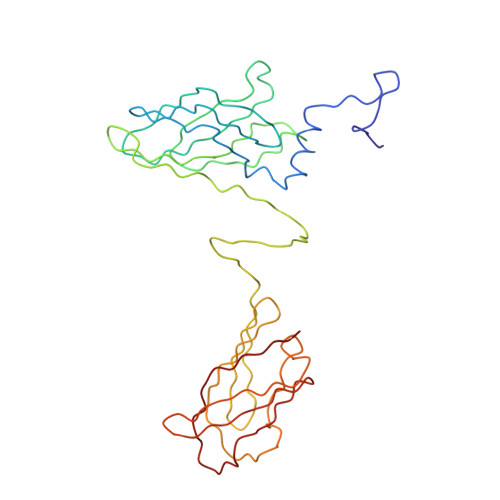
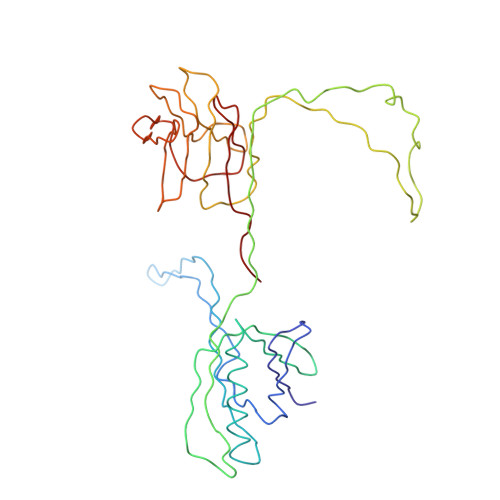
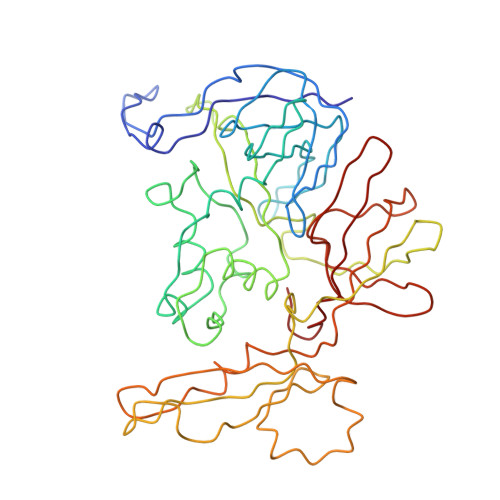
Structural Insights into Lactococcal Siphophage p2 Baseplate Activation Mechanism.
Spinelli, S., Tremblay, D., Moineau, S., Cambillau, C., Goulet, A.(2020) Viruses 12
- PubMed: 32796652 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.3390/v12080878
- Primary Citation Related Structures:
6ZIG, 6ZIH, 6ZJJ - PubMed Abstract:
Virulent phages infecting L. lactis , an industry-relevant bacterium, pose a significant risk to the quality of the fermented milk products. Phages of the Skunavirus genus are by far the most isolated lactococcal phages in the cheese environments and phage p2 is the model siphophage for this viral genus. The baseplate of phage p2, which is used to recognize its host, was previously shown to display two conformations by X-ray crystallography, a rested state and an activated state ready to bind to the host. The baseplate became only activated and opened in the presence of Ca 2+ . However, such an activated state was not previously observed in the virion. Here, using nanobodies binding to the baseplate, we report on the negative staining electron microscopy structure of the activated form of the baseplate directly observed in the p2 virion, that is compatible with the activated baseplate crystal structure. Analyses of this new structure also established the presence of a second distal tail (Dit) hexamer as a component of the baseplate, the topology of which differs largely from the first one. We also observed an uncoupling between the baseplate activation and the tail tip protein (Tal) opening, suggesting an infection mechanism more complex than previously expected.
- Architecture et Fonction des Macromolécules Biologiques, Aix-Marseille Université, Campus de Luminy, 13288 Marseille CEDEX 09, France.
Organizational Affiliation: