Co-crystallization, nanoESI-MS and 19F NMR reveal dimer disturbing inhibitors and conformational changes at dimer contacts
Nguyen, A., Heine, A., Klebe, G.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Queuine tRNA-ribosyltransferase | 388 | Zymomonas mobilis subsp. mobilis ZM4 = ATCC 31821 | Mutation(s): 1 Gene Names: tgt, ZMO0363 EC: 2.4.2.29 | 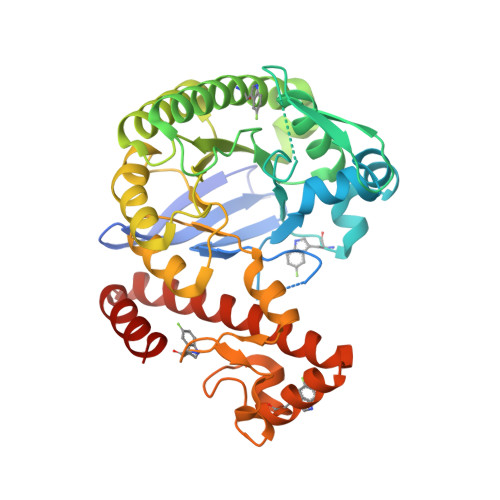 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P28720 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 726 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth A] | 6-azanyl-2-(methylamino)-4-[2-[(2~{R},3~{R},4~{S},5~{R},6~{S})-3,4,5,6-tetramethoxyoxan-2-yl]ethyl]-1,7-dihydroimidazo[4,5-g]quinazolin-8-one C21 H30 N6 O6 IXUIMEBGCTWFOR-ZIIYPAMZSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | B [auth A] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| FTR Query on FTR | A | L-PEPTIDE LINKING | C11 H11 F N2 O2 |  | TRP |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 85.083 | α = 90 |
| b = 64.98 | β = 93.776 |
| c = 71.374 | γ = 90 |
| Software Name | Purpose |
|---|---|
| Coot | model building |
| PHENIX | refinement |
| XDS | data reduction |
| XDS | data scaling |
| PHASER | phasing |