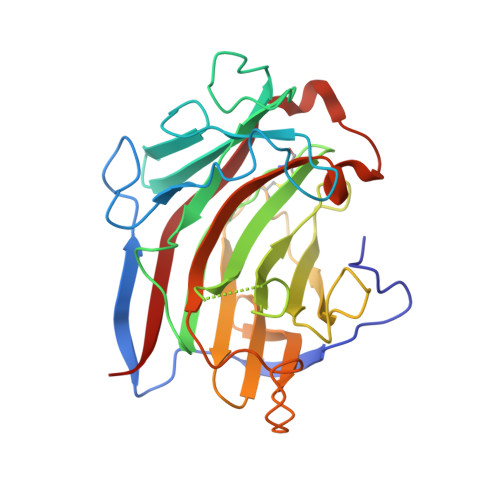
Insights into the dual cleavage activity of the GH16 laminarinase enzyme class on beta-1,3 and beta-1,4 glycosidic bonds.
Liberato, M.V., Teixeira Prates, E., Goncalves, T.A., Bernardes, A., Vilela, N., Fattori, J., Ematsu, G.C., Chinaglia, M., Machi Gomes, E.R., Migliorini Figueira, A.C., Damasio, A., Polikarpov, I., Skaf, M.S., Squina, F.M.(2021) J Biological Chem 296: 100385-100385
- PubMed: 33556371 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jbc.2021.100385
- Primary Citation Related Structures:
6XOF, 6XQF, 6XQG, 6XQH, 6XQL, 6XQM - PubMed Abstract:
Glycoside hydrolases (GHs) are involved in the degradation of a wide diversity of carbohydrates and present several biotechnological applications. Many GH families are composed of enzymes with a single well-defined specificity. In contrast, enzymes from the GH16 family can act on a range of different polysaccharides, including β-glucans and galactans. SCLam, a GH16 member derived from a soil metagenome, an endo-β-1,3(4)-glucanase (EC 3.2.1.6), can cleave both β-1,3 and β-1,4 glycosidic bonds in glucans, such as laminarin, barley β-glucan, and cello-oligosaccharides. A similar cleavage pattern was previously reported for other GH16 family members. However, the molecular mechanisms for this dual cleavage activity on (1,3)- and (1,4)-β-D-glycosidic bonds by laminarinases have not been elucidated. In this sense, we determined the X-ray structure of a presumably inactive form of SCLam cocrystallized with different oligosaccharides. The solved structures revealed general bound products that are formed owing to residual activities of hydrolysis and transglycosylation. Biochemical and biophysical analyses and molecular dynamics simulations help to rationalize differences in activity toward different substrates. Our results depicted a bulky aromatic residue near the catalytic site critical to select the preferable configuration of glycosidic bonds in the binding cleft. Altogether, these data contribute to understanding the structural basis of recognition and hydrolysis of β-1,3 and β-1,4 glycosidic linkages of the laminarinase enzyme class, which is valuable for future studies on the GH16 family members and applications related to biomass conversion into feedstocks and bioproducts.
- Laboratório Nacional de Ciência e Tecnologia do Bioetanol (CTBE), Centro Nacional de Pesquisa em Energia e Materiais (CNPEM), Campinas, São Paulo, Brazil; Programa de Processos Tecnológicos e Ambientais, Universidade de Sorocaba, Sorocaba, São Paulo, Brazil.
Organizational Affiliation: