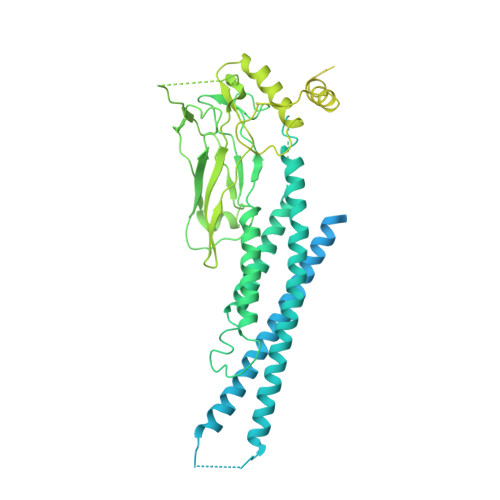
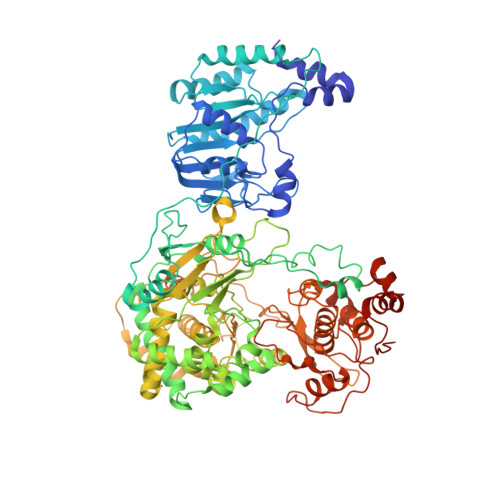
Structural basis for STAT2 suppression by flavivirus NS5.
Wang, B., Thurmond, S., Zhou, K., Sanchez-Aparicio, M.T., Fang, J., Lu, J., Gao, L., Ren, W., Cui, Y., Veit, E.C., Hong, H., Evans, M.J., O'Leary, S.E., Garcia-Sastre, A., Zhou, Z.H., Hai, R., Song, J.(2020) Nat Struct Mol Biol 27: 875-885
- PubMed: 32778820 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-020-0472-y
- Primary Citation Related Structures:
6UX2, 6WCZ - PubMed Abstract:
Suppressing cellular signal transducers of transcription 2 (STAT2) is a common strategy that viruses use to establish infections, yet the detailed mechanism remains elusive, owing to a lack of structural information about the viral-cellular complex involved. Here, we report the cryo-EM and crystal structures of human STAT2 (hSTAT2) in complex with the non-structural protein 5 (NS5) of Zika virus (ZIKV) and dengue virus (DENV), revealing two-pronged interactions between NS5 and hSTAT2. First, the NS5 methyltransferase and RNA-dependent RNA polymerase (RdRP) domains form a conserved interdomain cleft harboring the coiled-coil domain of hSTAT2, thus preventing association of hSTAT2 with interferon regulatory factor 9. Second, the NS5 RdRP domain also binds the amino-terminal domain of hSTAT2. Disruption of these ZIKV NS5-hSTAT2 interactions compromised NS5-mediated hSTAT2 degradation and interferon suppression, and viral infection under interferon-competent conditions. Taken together, these results clarify the mechanism underlying the functional antagonism of STAT2 by both ZIKV and DENV.
- Department of Biochemistry, University of California, Riverside, CA, USA.
Organizational Affiliation: