Structure of the human clamp loader bound to the sliding clamp: a further twist on AAA+ mechanism
Gaubitz, C., Liu, X., Magrino, J., Stone, N.P., Landeck, J., Hedglin, M., Kelch, B.A.(2020) bioRxiv
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
(2020) bioRxiv
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Replication factor C subunit 1 | 595 | Homo sapiens | Mutation(s): 0 Gene Names: RFC1, RFC140 | 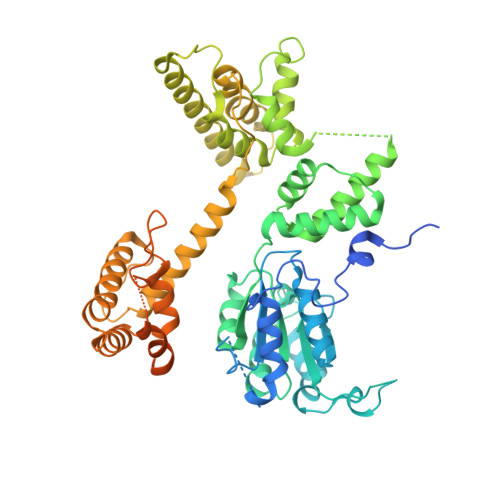 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P35251 GTEx: ENSG00000035928 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P35251 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Replication factor C subunit 2 | 354 | Homo sapiens | Mutation(s): 0 Gene Names: RFC2 | 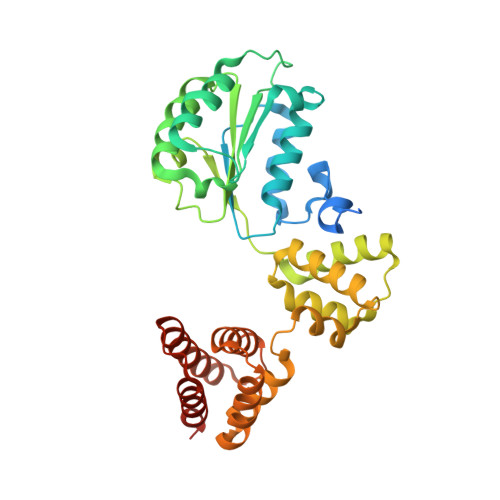 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P35250 GTEx: ENSG00000049541 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P35250 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Replication factor C subunit 5 | 340 | Homo sapiens | Mutation(s): 0 Gene Names: RFC5 | 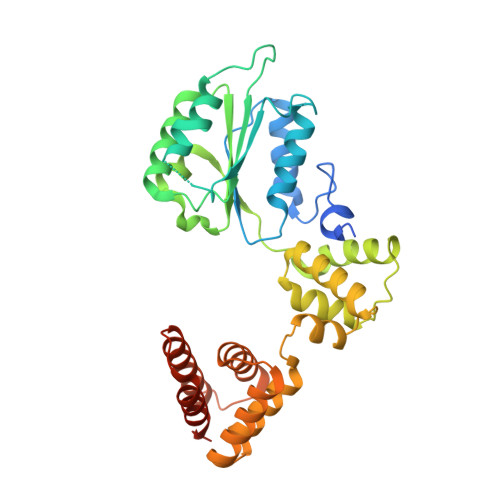 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P40937 GTEx: ENSG00000111445 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P40937 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Replication factor C subunit 4 | 363 | Homo sapiens | Mutation(s): 0 Gene Names: RFC4 | 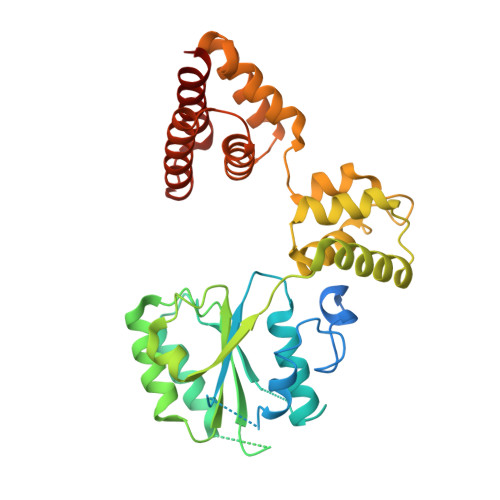 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P35249 GTEx: ENSG00000163918 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P35249 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Replication factor C subunit 3 | 356 | Homo sapiens | Mutation(s): 0 Gene Names: RFC3 |  | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P40938 GTEx: ENSG00000133119 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P40938 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 6 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Proliferating cell nuclear antigen | 261 | Homo sapiens | Mutation(s): 0 Gene Names: PCNA |  | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P12004 GTEx: ENSG00000132646 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P12004 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| AGS (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | J [auth A], L [auth B], N [auth C], P [auth D] | PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTER C10 H16 N5 O12 P3 S NLTUCYMLOPLUHL-KQYNXXCUSA-N |  | ||
| ADP (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | Q [auth E] | ADENOSINE-5'-DIPHOSPHATE C10 H15 N5 O10 P2 XTWYTFMLZFPYCI-KQYNXXCUSA-N |  | ||
| MG (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | I [auth A], K [auth B], M [auth C], O [auth D] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | |
| RECONSTRUCTION | RELION | 3.0.2 |
| Funding Organization | Location | Grant Number |
|---|---|---|
| American Cancer Society | United States | 440685 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | R01-GM127776 |
| Swiss National Science Foundation | Switzerland | 177859 |