Comprehensive structure-function characterization of DNMT3B and DNMT3A reveals distinctive de novo DNA methylation mechanisms
Gao, L.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA (cytosine-5)-methyltransferase 3B | 291 | Homo sapiens | Mutation(s): 1 Gene Names: DNMT3B EC: 2.1.1.37 | 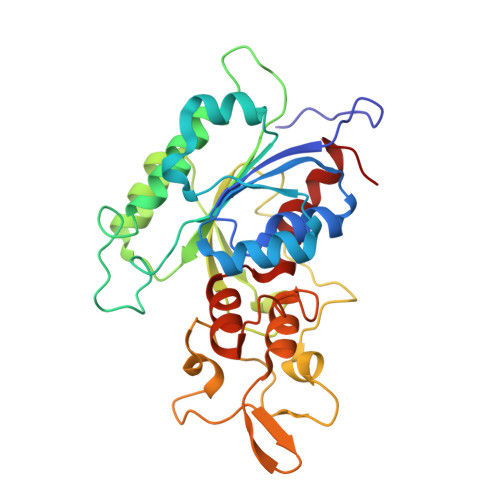 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9UBC3 GTEx: ENSG00000088305 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9UBC3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA (cytosine-5)-methyltransferase 3-like | 209 | Homo sapiens | Mutation(s): 0 Gene Names: DNMT3L |  | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9UJW3 GTEx: ENSG00000142182 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9UJW3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| CpGpT DNA (25-MER) | 25 | Homo sapiens | 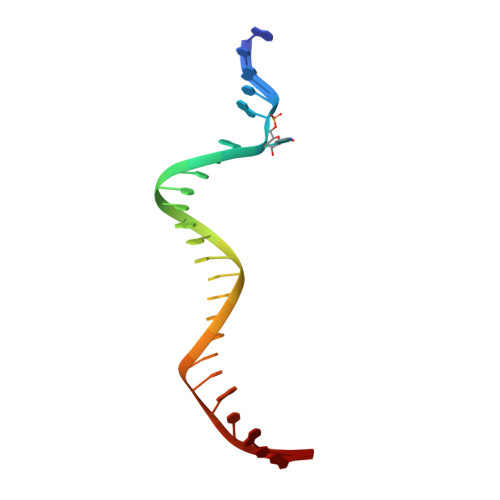 | |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SAH (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | H [auth A], J [auth D] | S-ADENOSYL-L-HOMOCYSTEINE C14 H20 N6 O5 S ZJUKTBDSGOFHSH-WFMPWKQPSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | G [auth A], I [auth D] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 191.506 | α = 90 |
| b = 191.506 | β = 90 |
| c = 49.597 | γ = 120 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Human Genome Research Institute (NIH/NHGRI) | United States | 1R35GM119721 |