Crystal Structures of Angiopoietin-Like 4s C-Terminal Domain (cANGPTL4) Reveal a Binding Site for Various Ligands
Tarver, C.L., Yuan, Q., Singhal, A.J., Ramaker, R., Cooper, S., Pusey, M.L.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Angiopoietin-related protein 4 | 216 | Homo sapiens | Mutation(s): 0 Gene Names: ANGPTL4, ARP4, HFARP, PGAR, PP1158, PSEC0166, UNQ171/PRO197 | 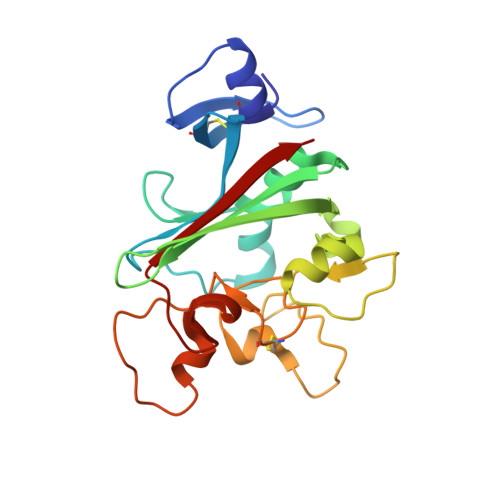 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9BY76 GTEx: ENSG00000167772 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9BY76 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| MYR Download:Ideal Coordinates CCD File | B [auth A] | MYRISTIC ACID C14 H28 O2 TUNFSRHWOTWDNC-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 132.496 | α = 90 |
| b = 135.632 | β = 90 |
| c = 39.596 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| AutoSol | phasing |
| PHENIX | model building |
| REFMAC | refinement |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Human Genome Research Institute (NIH/NHGRI) | United States | 5R42GM116283-03 |