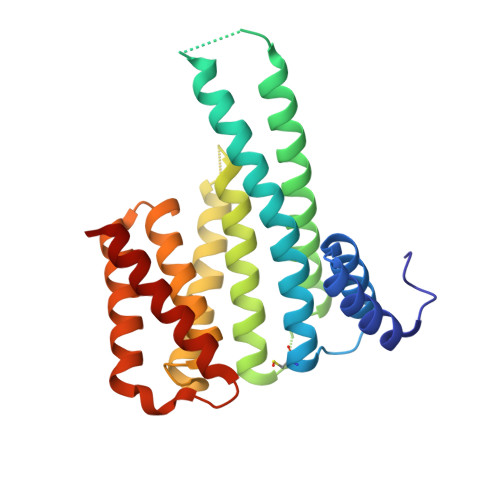
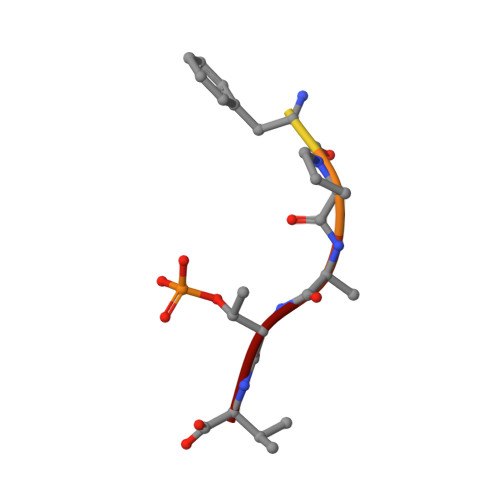
Design of Drug-Like Protein-Protein Interaction Stabilizers Guided By Chelation-Controlled Bioactive Conformation Stabilization.
Bosica, F., Andrei, S.A., Neves, J.F., Brandt, P., Gunnarsson, A., Landrieu, I., Ottmann, C., O'Mahony, G.(2020) Chemistry 26: 7131-7139
- PubMed: 32255539 Search on PubMed
- DOI: https://doi.org/10.1002/chem.202001608
- Primary Citation Related Structures:
6TJM, 6TL3 - PubMed Abstract:
Protein-protein interactions (PPIs) of 14-3-3 proteins are a model system for studying PPI stabilization. The complex natural product Fusicoccin A stabilizes many 14-3-3 PPIs but is not amenable for use in SAR studies, motivating the search for more drug-like chemical matter. However, drug-like 14-3-3 PPI stabilizers enabling such studies have remained elusive. An X-ray crystal structure of a PPI in complex with an extremely low potency stabilizer uncovered an unexpected non-protein interacting, ligand-chelated Mg 2+ leading to the discovery of metal-ion-dependent 14-3-3 PPI stabilization potency. This originates from a novel chelation-controlled bioactive conformation stabilization effect. Metal chelation has been associated with pan-assay interference compounds (PAINS) and frequent hitter behavior, but chelation can evidently also lead to true potency gains and find use as a medicinal chemistry strategy to guide compound optimization. To demonstrate this, we exploited the effect to design the first potent, selective, and drug-like 14-3-3 PPI stabilizers.
- Research and Early Development, Cardiovascular, Renal and Metabolism, BioPharmaceuticals R&D, AstraZeneca, Pepparedsleden 1, 43183, Mölndal, Sweden.
Organizational Affiliation: