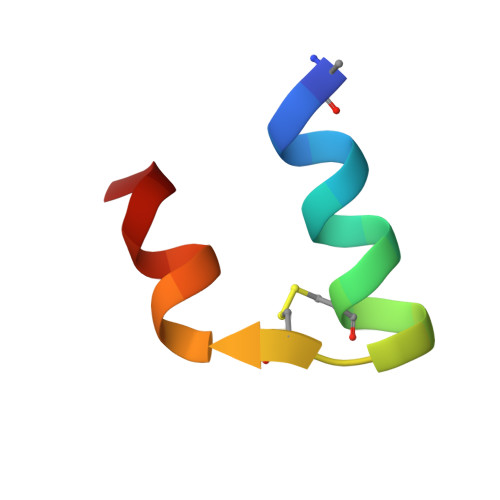
X-ray structure of human relaxin at 1.5 A. Comparison to insulin and implications for receptor binding determinants.
Eigenbrot, C., Randal, M., Quan, C., Burnier, J., O'Connell, L., Rinderknecht, E., Kossiakoff, A.A.(1991) J Mol Biology 221: 15-21
- PubMed: 1656049 Search on PubMed
- Primary Citation Related Structures:
6RLX - PubMed Abstract:
The X-ray crystal structure of relaxin at 1.5 A resolution is reported for the physiologically active form of the human hormone. Relaxin is a small, two-chain polypeptide that is a member of the protein hormone family that also includes insulin and the insulin-like growth factors IGF-I and IGF-II. These hormones have biologically diverse activities but are structurally similar, sharing a distinctive pattern of cysteine and glycine residues. The predicted structural homology of relaxin to insulin is confirmed by this structural analysis; however, there are significant differences in the terminal regions of the b-chain. Although relaxin, like insulin, crystallizes as a dimer, the orientation of the molecules in the respective dimers is completely different. The region of the relaxin molecule proposed to be involved in receptor binding is part of the dimer interface, suggesting that some of the other residues contained in the dimer contact surface might be receptor binding determinants as well. The proposed receptor binding determinants for insulin likewise include residues at its dimer interface. However, because the dimer contacts of relaxin and insulin are quite different, it appears that these two structurally related hormones have evolved somewhat dissimilar mechanisms for receptor binding.
- Protein Engineering, Bioorganic Chemistry Department, Genentech, Inc., South San Francisco, CA 94080.
Organizational Affiliation: